- Posts: 210
 Periodic dimerization and backward.py mapping
Periodic dimerization and backward.py mapping
- Robin
-
 Topic Author
Topic Author
- Visitor
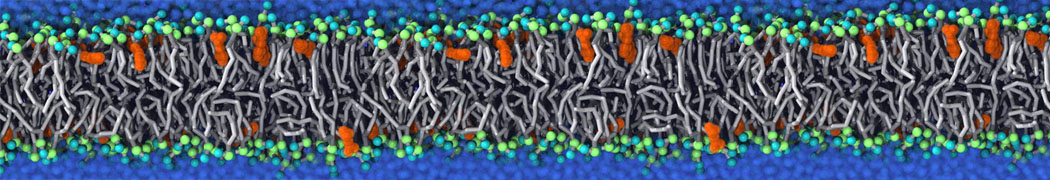
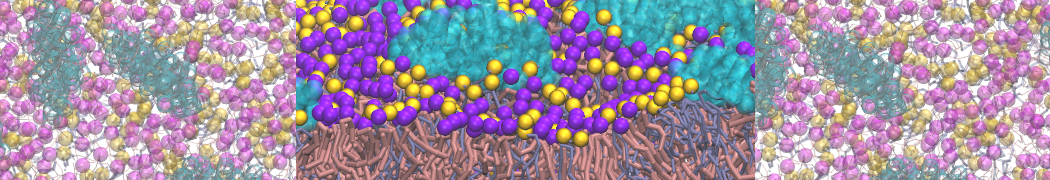
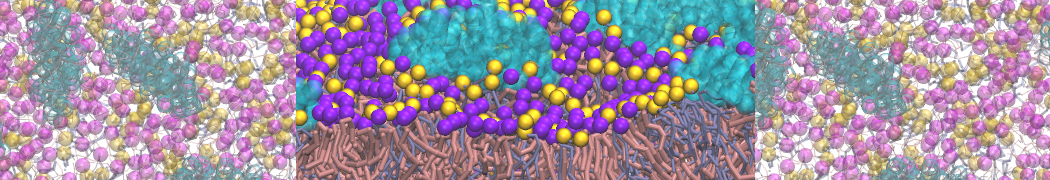
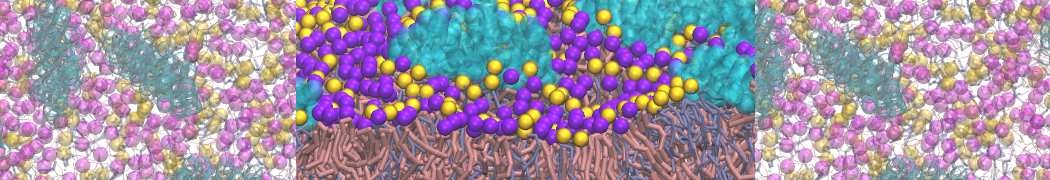
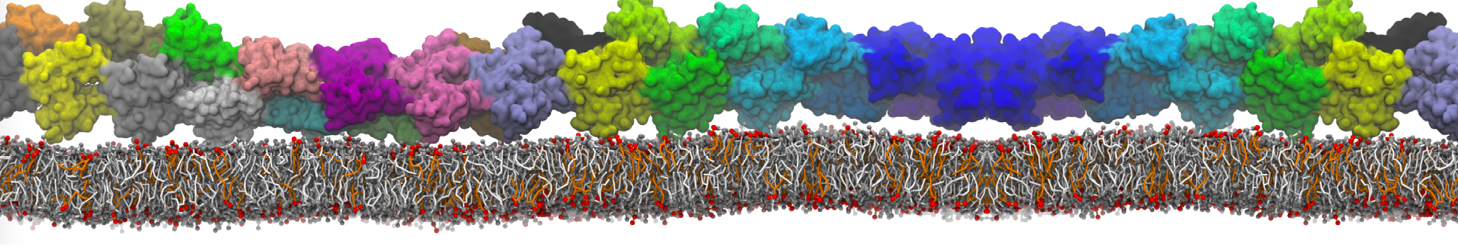
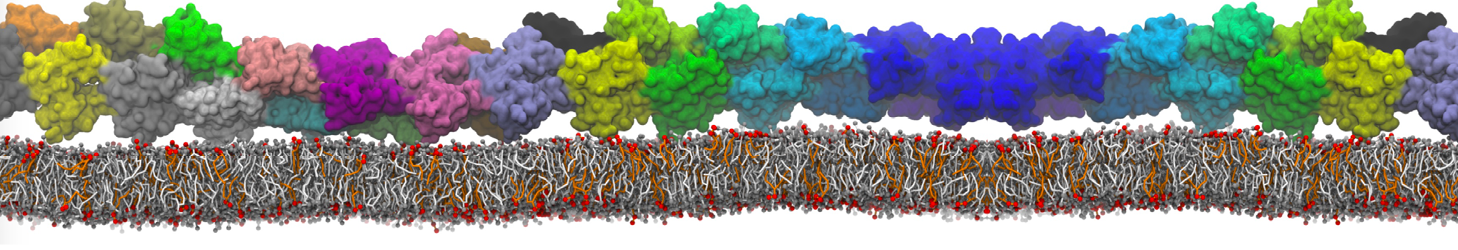
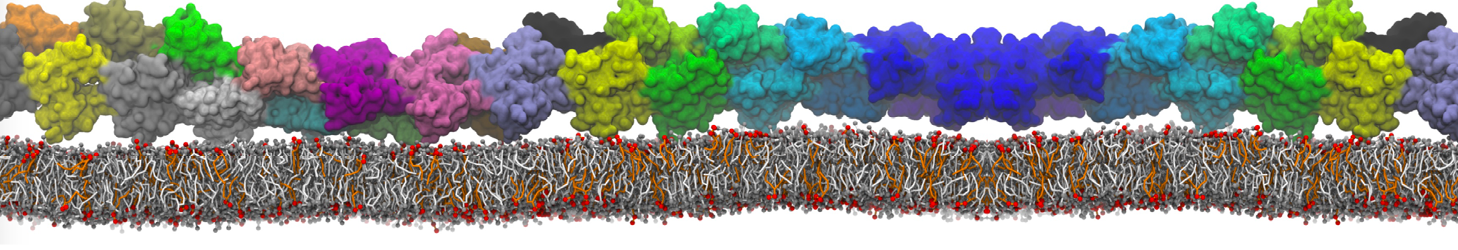
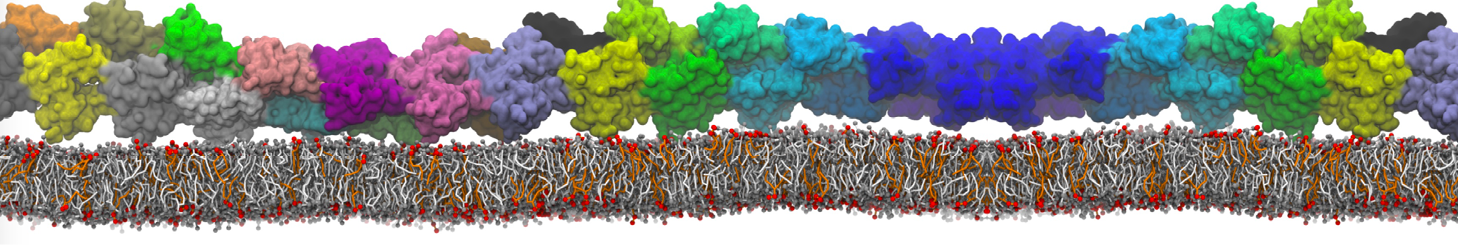
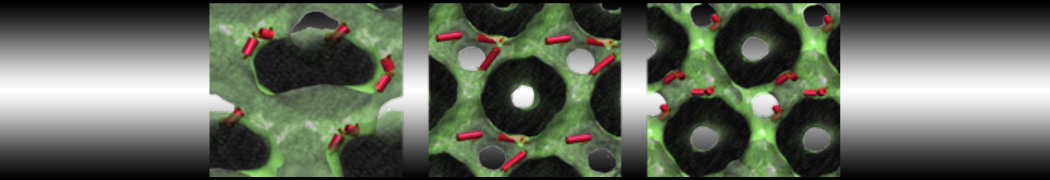
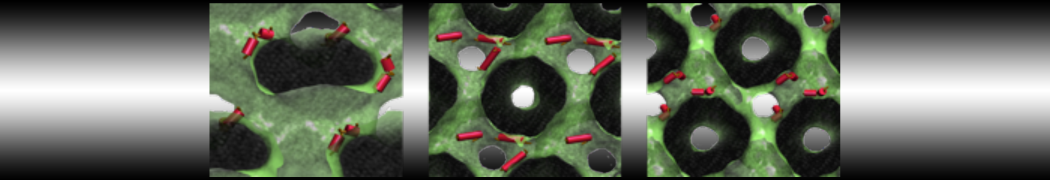
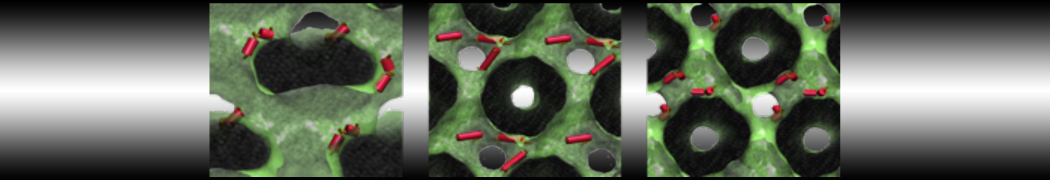
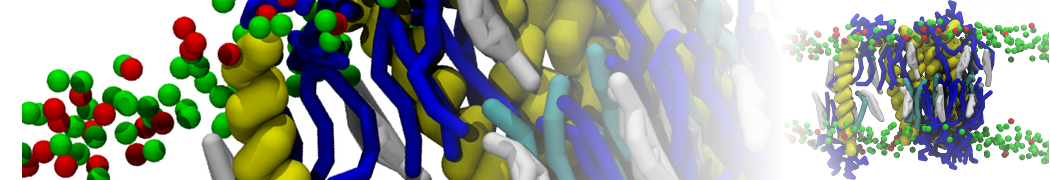
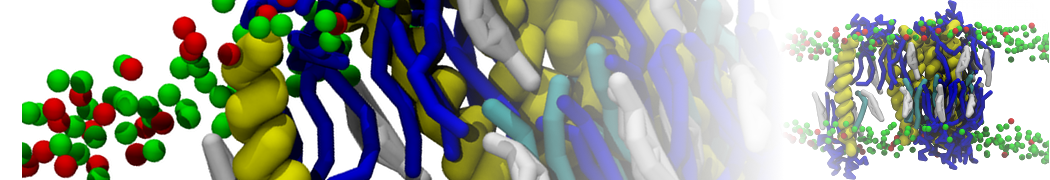
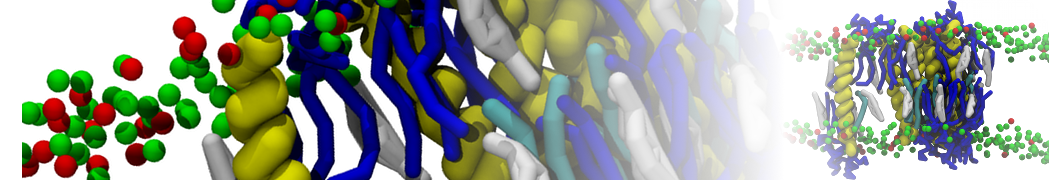
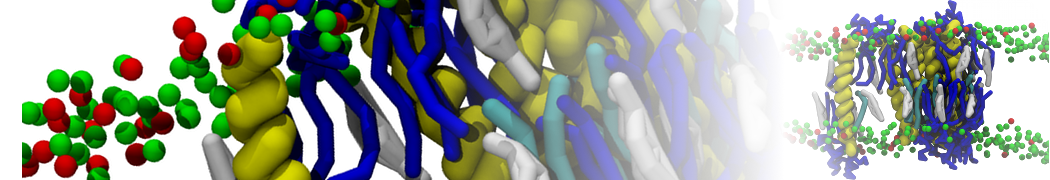
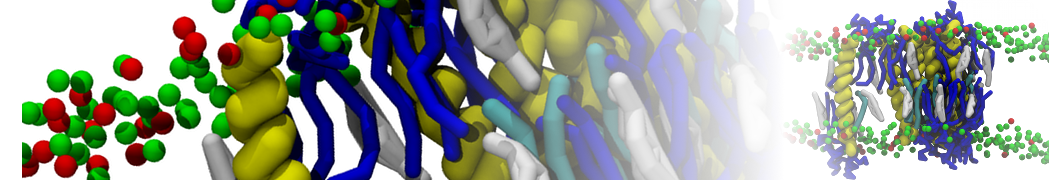
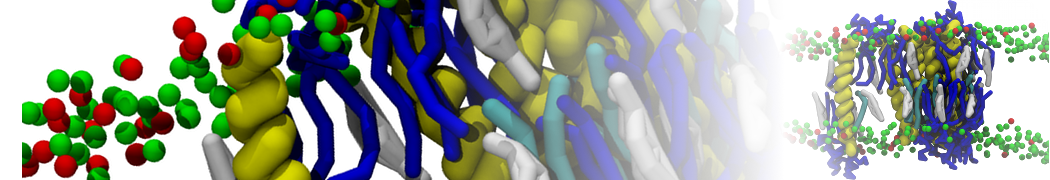
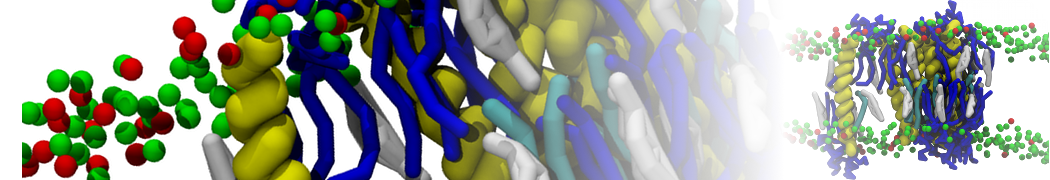
I am currently studying dimerization interface of 2 proteins in martini representation, and trying to backmap the system (dimer+membrane) in order to study it in all atom later.
My first problem concern directly one of my dimers which dimerized at the extremity of the box. I am pretty sure the dimerization happened since I can see the dimer by using periodic images. But evidently, when I try to locate interacting residue (this script is pretty usefull for that purpose: www.ks.uiuc.edu/Research/vmd/mailing_lis...5681/newcontacts.tcl ), the script doesn't see the interacting image.
I tried several combination of gmx trjconv to make one of the protomer the center:
gmx trjconv -f half101prot10ns.trr -s dimer.gro -o recentered10ns.trr -b 0 -n mudelta.ndx -center -pbc whole -boxcenter
But no success, my protomers stay at the opposite periodic "wall".
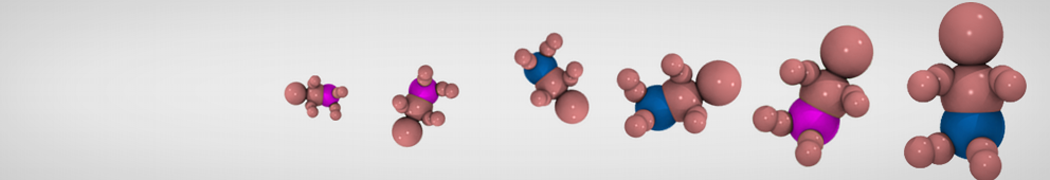
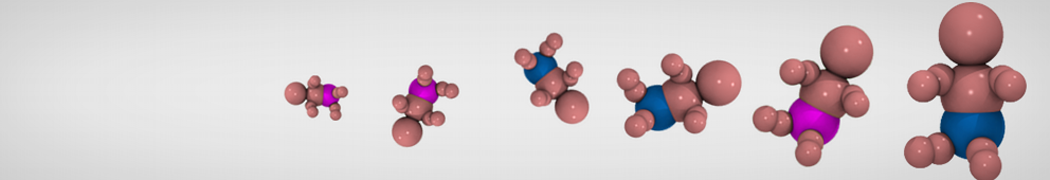
My second problem concern backward.py handling of the POPC lipid:
want: H91
have:
Bailing out...
I know this problem has already been adressed :
flaviyan wrote: Hi Chris,
You are using the Martini lipid rule 2.0 for the POPC [12 beads] (which is the case in the latest insane script) but you are using the Mapping scheme according to the old lipid rules [13 beads]. so the backward is throwing an error. Either update the Mapping to match the new bead description or use the following insane command:
./insane.py -o cg.gro -p cg.top -l POPC.o:1 -x 10 -y 10 -z 10
So I changed the POPC mapping using this page: cgmartini.nl/index.php/force-field-param...ml?dir=PC&lipid=POPC .
The error is still the same despite using the 2.0 martini rule. Do you have any other idea of what could cause this problem?
Thank you for reading :).
Robin
Please Log in or Create an account to join the conversation.
- peterkroon
-

- Offline
- Gold Boarder
Please Log in or Create an account to join the conversation.
- Robin
-
 Topic Author
Topic Author
- Visitor
Finally, It seems the following command did the trick (much more simple than I though):
gmx trjconv -f halfsystem201.trr -s prod1.tpr -pbc cluster -o halfprot102.trr
My dimer is finally whole!
Still no solution for backward.py.
Please Log in or Create an account to join the conversation.
- Robin
-
 Topic Author
Topic Author
- Visitor
[('H45', None), ('C47', None), ('C1', (1.0376332995838211, 5.433177095506531, 4.253398502591266)), ('O2', None)]
Not all positions defined for [trans] operation:
[('H46', None), ('C47', None), ('C1', (1.0376332995838211, 5.433177095506531, 4.253398502591266)), ('O2', None)]
Not all positions defined for [trans] operation:
[('C44', None), ('C47', None), ('C1', (1.594848688419134, 8.273678175555368, 4.4328742475019665)), ('O2', None)]
I think this problem is linked to the POPC mapping but I don't understand really how it works.
Anyway, It doesn't seem to be a critical error since it continue to minimization until this error arise:
Fatal error:
Atomtype SP1 not found
I know it seems to be a ELnedyn atom (representation i'm using) but in my martini file used for backmapping, trp only got the BB, SC1, SC2, SC3 and SC4 types. Of course, this atome doesn't appear in the 0-backward.gro file either. Does it means that gmx or backward are expecting the SP1 atometype that doesn't exist in my files?
Update: it seems backward.py doesn't FG the cholesterol residue in my file. Pretty strange since it does for POPC and all the residue of my protein: the .map file of chol is in the same folder as the others.
Please Log in or Create an account to join the conversation.
- tsjerk
-

- Offline
- Expert Boarder
- Posts: 103
Please Log in or Create an account to join the conversation.
- Robin
-
 Topic Author
Topic Author
- Visitor
I tried to make an chol.itp file from one standard cholesterol in all-atom, and from the charmm 36 forcefield. Problem is the name and atom numbering doesn't seem to be the same in chol.charmm36.map and the charmm36 forcefield. Does anyone know from what all atom topology the cholesterol mapping was done? Only solution I see for my problem is replacing one by one the name of the 74 cholesterol atoms in the map file.
Moreover I think I spotted an error in the chol.charmm36.map:
[out]
C52 C50 C56 C24
H53 C50 C56 C24
H54 C50 C56 C24
H44 C50 C56 C24
The H44 atom doesn't exist in the [ atom ] section, I guess H44=C44?
Please Log in or Create an account to join the conversation.