- Posts: 3
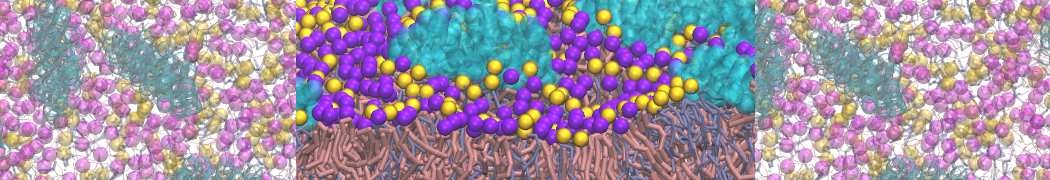
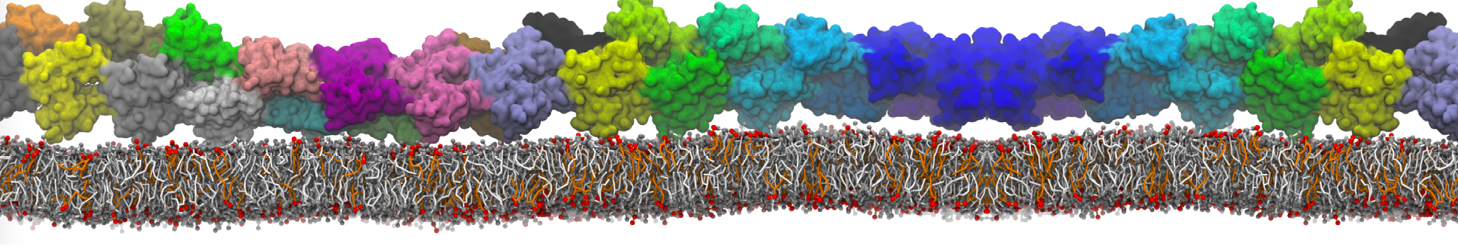
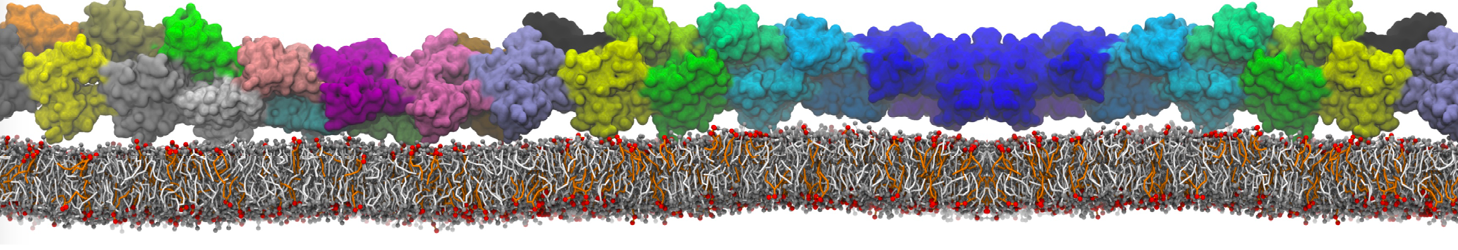
 protein-membrane (POPG) interaction
protein-membrane (POPG) interaction
- busato
-
 Topic Author
Topic Author
- Offline
- Fresh Boarder
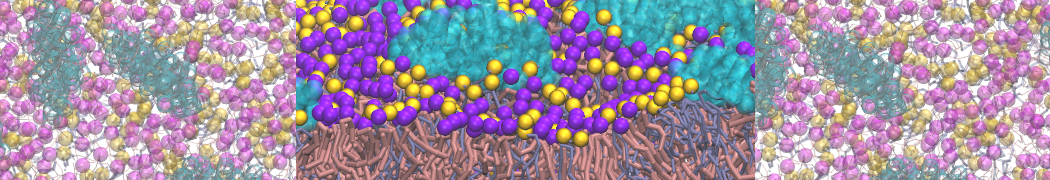
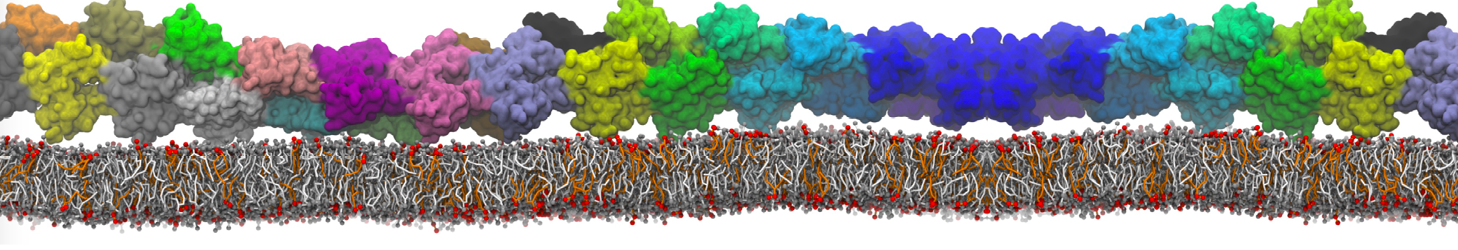
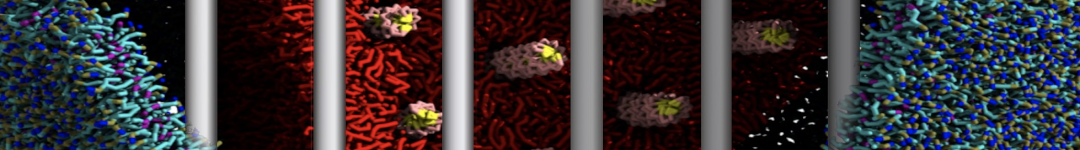
I am a new user of Martini ff, I simulated the interaction between a protein and membrane.
First, I am interested to know which aminoacids are involved in the interaction with the membrane along my trajectory. (for example which aa are within of 0.6 nm from the surface of the membrane).
Are there any scripts or tools to perform this kind of analysis?
Second, I would like to know the limit of distance of a acceptable salt bridge interaction in Martini ff.
I really appreciate your help.
Thank you,
Mirko
Please Log in or Create an account to join the conversation.
- Clement
-

- Offline
- Admin
- Posts: 211
Welcome Mirko!I am a new user of Martini ff, I simulated the interaction between a protein and membrane.
I'd check g_select, included in the Gromacs package. Syntax very comprehensible.First, I am interested to know which aminoacids are involved in the interaction with the membrane along my trajectory. (for example which aa are within of 0.6 nm from the surface of the membrane).
Are there any scripts or tools to perform this kind of analysis?
For that one... If by salt bridge you mean hydrogen bonding, remember you're dealing with CG here, and not such things exist... What do you want to check/quantify/measure exactly?Second, I would like to know the limit of distance of a acceptable salt bridge interaction in Martini ff.
Please Log in or Create an account to join the conversation.
- busato
-
 Topic Author
Topic Author
- Offline
- Fresh Boarder
- Posts: 3
Yes I checked g_selected and I also found a command g_mindist.
Yes, you are right, Infact now I have performed a map of contact between protein (aa) and membrane (cutoff 0.4 nm).
1)I would like to find a way to quantify and understand the possible interactions and the type of interaction.
2)What interactions can I study in Martini ff? and What rules these interactions have to follow (distances, angles ..)?
Thank you very much,
Mirko
Please Log in or Create an account to join the conversation.
- Clement
-

- Offline
- Admin
- Posts: 211
0.4 nm seems a bit too short to me... Remember Martini beads have an effective radius of 0.47 nm in average. I'd increase your cutoff to 0.7 nm at least, if not slightly more.[...] map of contact between protein (aa) and membrane (cutoff 0.4 nm).
Well... There I'd need more information about what you mean by that. Martini defined non-bonded interactions are van der Waals and electrostatic, but hydrophobic interactions can be observed equally. What do you want to analyze exactly?[...] quantify and understand the possible interactions and the type of interaction.
There again I need more precisions. Isn't this question a bit similar to your previous one? At least I understand it this way.What interactions can I study in Martini ff? and What rules these interactions have to follow (distances, angles ..)?
EDIT: I guess you want to check things in a similar way as hydrogen bonds or something. Directivity, distance, angle, ... defining if interaction there is. I think you have to define your proper rules in Martini if you really need them. But remember you're dealing with a CG FF, and interactions loose most of their directivity as compared to AA simulations. You have to rethink the way of analyzing things, but it's more a simplification.
Please Log in or Create an account to join the conversation.
- busato
-
 Topic Author
Topic Author
- Offline
- Fresh Boarder
- Posts: 3
I used in my analysis 0.7 nm (cut-off for the distance between protein and membrane) as you suggested me.
Can you suggest me some papers which I can find and read in detail about the Martini beads have an effective radius of 0.47 nm in average?
Thank you very much,
Mirko
Please Log in or Create an account to join the conversation.
- helgi
-

- Offline
- Administrator
- Posts: 54
S.J. Marrink, H.J. Risselada, S. Yefimov, D.P. Tieleman, A.H. de Vries. The MARTINI forcefield: coarse grained model for biomolecular simulations. JPC-B, 111:7812-7824, 2007.
S.J. Marrink, A.H. de Vries, A.E. Mark. Coarse grained model for semi-quantitative lipid simulations. JPC-B, 108:750-760, 2004
Cheers,
- Helgi
Please Log in or Create an account to join the conversation.