- Posts: 5
 Counter ions clumping together during production run
Counter ions clumping together during production run
- Anna Stephens
-
 Topic Author
Topic Author
- Offline
- Fresh Boarder
Less
More
1 year 11 months ago #9466
by Anna Stephens
Counter ions clumping together during production run was created by Anna Stephens
Hey,
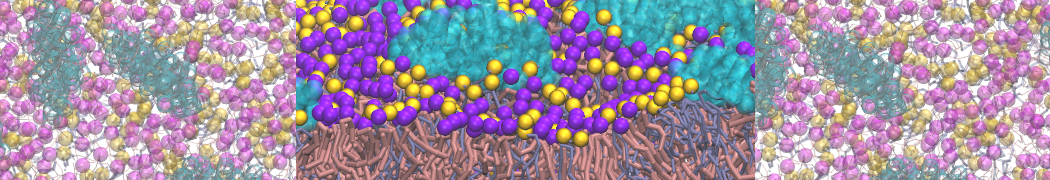
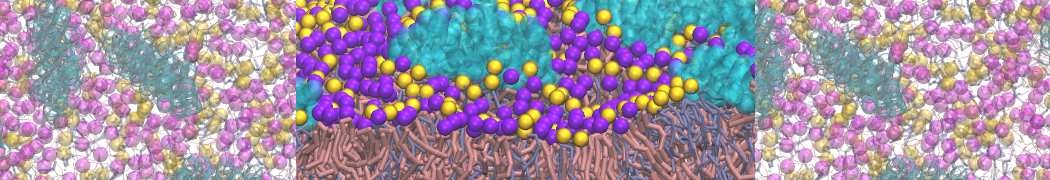
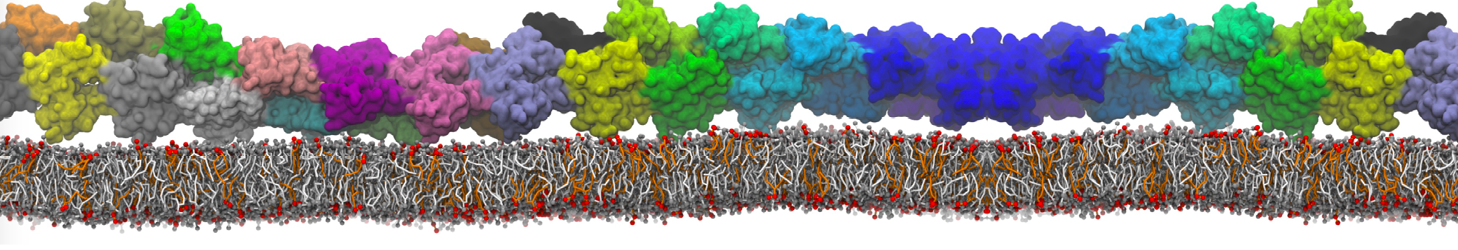
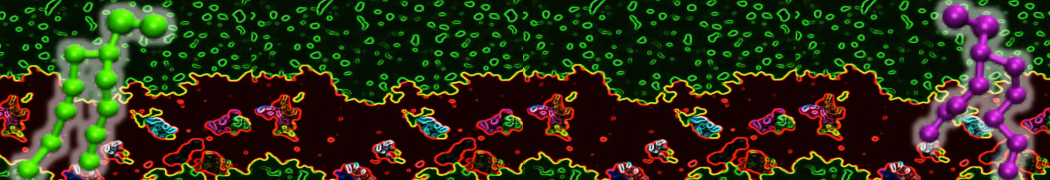
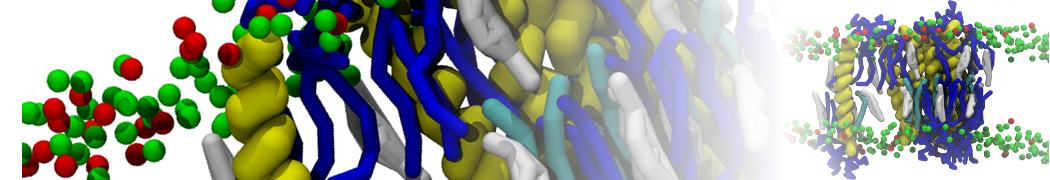
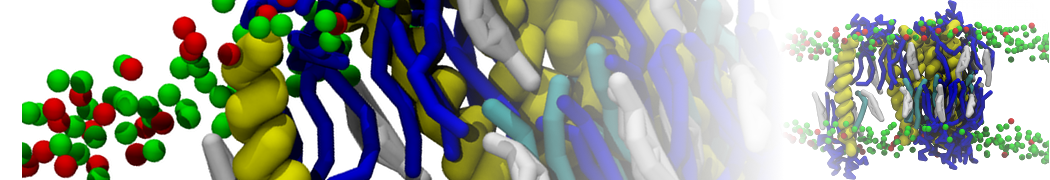
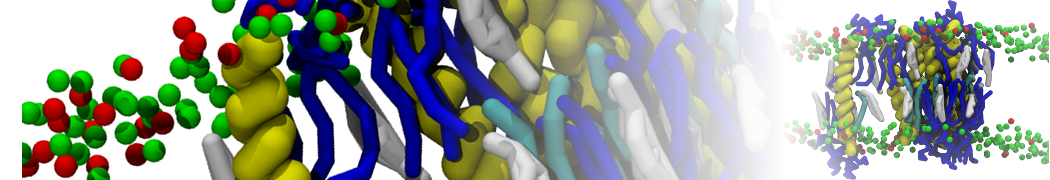
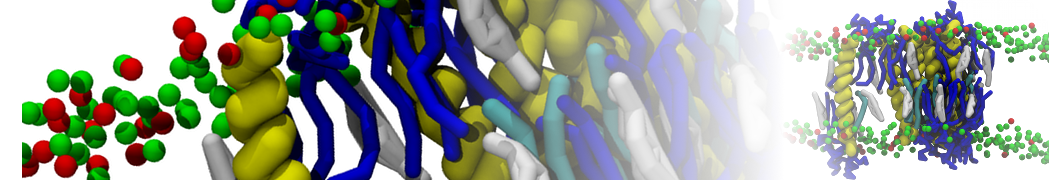
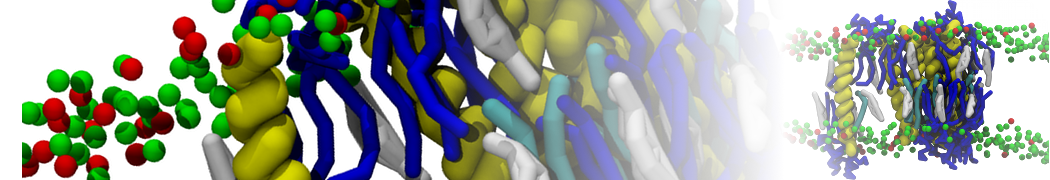
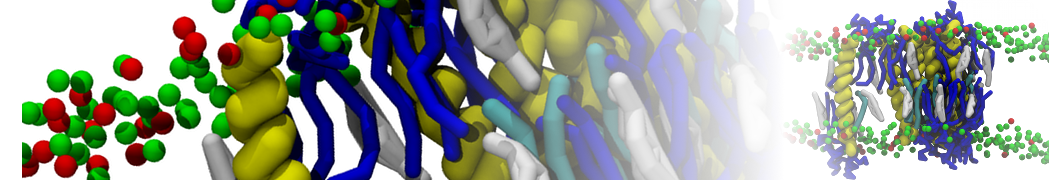
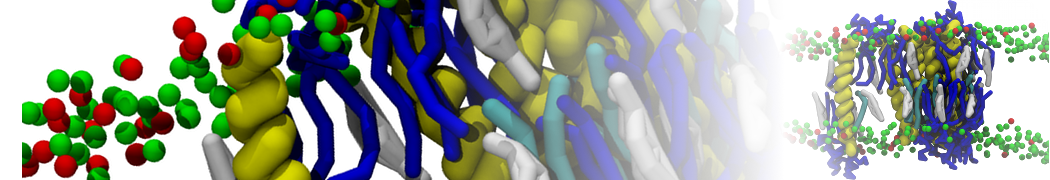
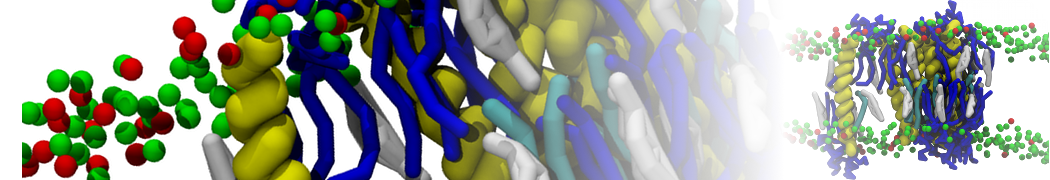
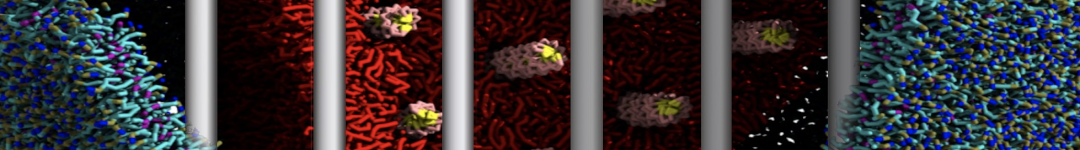
I'm running a 50 ns production simulation of a new CG peptide. During the production run all ions clump together in a string-like fashion then cluster together into large lumps in the system and around my peptide - is there a way I can stop this happening? They are NA and CL ions :)
Thanks!
Anna
I'm running a 50 ns production simulation of a new CG peptide. During the production run all ions clump together in a string-like fashion then cluster together into large lumps in the system and around my peptide - is there a way I can stop this happening? They are NA and CL ions :)
Thanks!
Anna
Please Log in or Create an account to join the conversation.
- vainikka
-

- Offline
- Admin
Less
More
- Posts: 91
1 year 11 months ago #9468
by vainikka
Replied by vainikka on topic Counter ions clumping together during production run
Hi,
I can think of two places to look for possible issues, with the first being the mdp settings. Are you using the standard settings for Martini?
The second possibility has to do with the peptide parameters - you mentioned that it is a new peptide, what bead types does it consist of?
I can think of two places to look for possible issues, with the first being the mdp settings. Are you using the standard settings for Martini?
The second possibility has to do with the peptide parameters - you mentioned that it is a new peptide, what bead types does it consist of?
Please Log in or Create an account to join the conversation.
- Anna Stephens
-
 Topic Author
Topic Author
- Offline
- Fresh Boarder
Less
More
- Posts: 5
1 year 11 months ago #9470
by Anna Stephens
Replied by Anna Stephens on topic Counter ions clumping together during production run
Hey,
My peptide consists of Serine, Lysine, Glycine and Isoleucine beads :)
The bead types are in the .gro file here:
1GLY BB 1 5.500 5.970 8.457 0.0321 -0.1889 -0.1381
2ILE BB 2 5.539 6.248 8.433 -0.0441 0.1175 0.2417
2ILE SC1 3 5.291 6.428 8.382 0.0048 0.0187 -0.3530
3ILE BB 4 5.821 6.380 8.433 -0.5382 -0.0295 -0.2408
3ILE SC1 5 5.785 6.682 8.374 -0.1138 0.0255 -0.2124
4LYS BB 6 6.129 6.322 8.335 -0.4419 0.1299 0.1312
4LYS SC1 7 6.338 6.170 8.517 0.2437 -0.0474 -0.0635
4LYS SC2 8 6.401 5.981 8.708 0.0359 0.1261 0.1606
5SER BB 9 6.088 6.204 8.059 -0.1296 -0.0241 0.1312
5SER SC1 10 5.873 6.046 8.046 -0.2040 0.2811 0.1788
6ILE BB 11 6.413 6.154 7.931 -0.0365 0.0844 -0.0531
6ILE SC1 12 6.456 6.402 7.750 -0.0853 0.0600 -0.0976
7ILE BB 13 6.578 5.918 7.839 0.3101 0.1128 -0.0811
7ILE SC1 14 6.568 5.972 7.534 0.1234 -0.0627 -0.1056
8LYS BB 15 6.718 5.608 7.922 0.0321 0.0824 -0.1903
8LYS SC1 16 6.457 5.392 7.987 -0.0138 0.1463 -0.0142
8LYS SC2 17 6.332 5.296 8.139 0.0954 -0.1162 0.0266
9SER BB 18 6.992 5.441 7.970 -0.3956 0.3567 -0.2319
9SER SC1 19 6.961 5.203 7.877 0.0884 -0.0812 -0.0177
10ILE BB 20 7.279 5.491 8.193 0.1563 -0.2357 0.2483
10ILE SC1 21 7.486 5.322 8.037 -0.0443 -0.4676 0.2354
11ILE BB 22 7.386 5.781 8.482 0.1303 -0.1111 -0.3149
11ILE SC1 23 7.204 6.010 8.379 0.0793 -0.0047 0.0102
12LYS BB 24 7.265 5.625 8.796 0.0478 0.0462 -0.0177
12LYS SC1 25 7.082 5.399 8.693 0.2565 -0.0043 -0.2232
12LYS SC2 26 6.907 5.199 8.802 0.2128 0.2818 0.2625
13SER BB 27 7.105 5.634 9.109 -0.0574 -0.2658 -0.1664
13SER SC1 28 7.158 5.839 9.263 0.0154 0.1371 0.0659
14ILE BB 29 6.843 5.390 9.250 0.1102 0.0637 -0.1556
14ILE SC1 30 7.037 5.237 9.438 0.1397 0.1830 -0.0906
The .mdp out file is the example one suggested in the tutorials below:
define = -DPOSRES
; RUN CONTROL PARAMETERS
integrator = md
; Start time and timestep in ps
tinit = 0
dt = 0.001
nsteps = 125000
; For exact run continuation or redoing part of a run
init-step = 0
; Part index is updated automatically on checkpointing (keeps files separate)
simulation-part = 1
; mode for center of mass motion removal
comm_mode = linear
; number of steps for center of mass motion removal
nstcomm = 100
; group(s) for center of mass motion removal
comm_grps = Protein W_NA+_CL-
; LANGEVIN DYNAMICS OPTIONS
; Friction coefficient (amu/ps) and random seed
bd-fric = 0
ld-seed = -1
; ENERGY MINIMIZATION OPTIONS
; Force tolerance and initial step-size
emtol = 10
emstep = 0.01
; Max number of iterations in relax-shells
niter = 20
; Step size (ps^2) for minimization of flexible constraints
fcstep = 0
; Frequency of steepest descents steps when doing CG
nstcgsteep = 1000
nbfgscorr = 10
; TEST PARTICLE INSERTION OPTIONS
rtpi = 0.05
; OUTPUT CONTROL OPTIONS
; Output frequency for coords (x), velocities (v) and forces (f)
nstxout = 0
nstvout = 5000
nstfout = 5000
; Output frequency for energies to log file and energy file
nstlog = 1000
nstcalcenergy = 100
nstenergy = 1000
; Output frequency and precision for .xtc file
nstxout-compressed = 5000
compressed-x-precision = 1000
; This selects the subset of atoms for the compressed
; trajectory file. You can select multiple groups. By
; default, all atoms will be written.
compressed-x-grps =
; Selection of energy groups
energygrps =
; NEIGHBORSEARCHING PARAMETERS
; cut-off scheme (Verlet: particle based cut-offs)
cutoff-scheme = Verlet
; nblist update frequency
nstlist = 20
; Periodic boundary conditions: xyz, no, xy
pbc = xyz
periodic-molecules = no
; Allowed energy error due to the Verlet buffer in kJ/mol/ps per atom,
; a value of -1 means: use rlist
verlet-buffer-tolerance = 0.005
; nblist cut-off
rlist = 1.2
; long-range cut-off for switched potentials
; OPTIONS FOR ELECTROSTATICS AND VDW
; Method for doing electrostatics
coulombtype = PME
coulomb-modifier = Potential-shift-Verlet
rcoulomb-switch = 0
rcoulomb = 1.2
; Relative dielectric constant for the medium and the reaction field
epsilon-r = 1
epsilon-rf = 0
; Method for doing Van der Waals
vdwtype = Cut-off
vdw-modifier = Force-switch
; cut-off lengths
rvdw_switch = 1.0
rvdw = 1.2
; Apply long range dispersion corrections for Energy and Pressure
DispCorr = No
; Extension of the potential lookup tables beyond the cut-off
table-extension = 1
; Separate tables between energy group pairs
energygrp-table =
; Spacing for the PME/PPPM FFT grid
fourierspacing = 0.12
; FFT grid size, when a value is 0 fourierspacing will be used
fourier-nx = 0
fourier-ny = 0
fourier-nz = 0
; EWALD/PME/PPPM parameters
pme-order = 4
ewald-rtol = 1e-05
ewald-rtol-lj = 0.001
lj-pme-comb-rule = Geometric
ewald-geometry = 3d
epsilon-surface = 0
implicit-solvent = no
; OPTIONS FOR WEAK COUPLING ALGORITHMS
; Temperature coupling
tcoupl = V-rescale
nsttcouple = -1
nh-chain-length = 10
print-nose-hoover-chain-variables = no
; Groups to couple separately
tc_grps = Protein W_NA+_CL-
; Time constant (ps) and reference temperature (K)
tau_t = 1.0 1.0
ref_t = 303.15 303.15
; pressure coupling
pcoupl = Berendsen
pcoupltype = Isotropic
nstpcouple = -1
; Time constant (ps), compressibility (1/bar) and reference P (bar)
tau_p = 5.0
compressibility = 4.5e-5 4.5e-5
ref_p = 1.0 1.0
; Scaling of reference coordinates, No, All or COM
refcoord_scaling = com
; OPTIONS FOR QMMM calculations
QMMM = no
; Groups treated Quantum Mechanically
QMMM-grps =
; QM method
QMmethod =
; QMMM scheme
QMMMscheme = normal
; QM basisset
QMbasis =
; QM charge
QMcharge =
; QM multiplicity
QMmult =
; Surface Hopping
SH =
; CAS space options
CASorbitals =
CASelectrons =
SAon =
SAoff =
SAsteps =
; Scale factor for MM charges
MMChargeScaleFactor = 1
; SIMULATED ANNEALING
; Type of annealing for each temperature group (no/single/periodic)
annealing =
; Number of time points to use for specifying annealing in each group
annealing-npoints =
; List of times at the annealing points for each group
annealing-time =
; Temp. at each annealing point, for each group.
annealing-temp =
; GENERATE VELOCITIES FOR STARTUP RUN
gen-vel = no
gen-temp = 300
gen-seed = -1
; OPTIONS FOR BONDS
constraints = h-bonds
; Type of constraint algorithm
constraint_algorithm = LINCS
; Do not constrain the start configuration
continuation = yes
; Use successive overrelaxation to reduce the number of shake iterations
Shake-SOR = no
; Relative tolerance of shake
shake-tol = 0.0001
; Highest order in the expansion of the constraint coupling matrix
lincs-order = 4
; Number of iterations in the final step of LINCS. 1 is fine for
; normal simulations, but use 2 to conserve energy in NVE runs.
; For energy minimization with constraints it should be 4 to 8.
lincs-iter = 1
; Lincs will write a warning to the stderr if in one step a bond
; rotates over more degrees than
lincs-warnangle = 30
; Convert harmonic bonds to morse potentials
morse = no
; ENERGY GROUP EXCLUSIONS
; Pairs of energy groups for which all non-bonded interactions are excluded
energygrp-excl =
; WALLS
; Number of walls, type, atom types, densities and box-z scale factor for Ewald
nwall = 0
wall-type = 9-3
wall-r-linpot = -1
wall-atomtype =
wall-density =
wall-ewald-zfac = 3
; COM PULLING
pull = no
; AWH biasing
awh = no
; ENFORCED ROTATION
; Enforced rotation: No or Yes
rotation = no
; Group to display and/or manipulate in interactive MD session
IMD-group =
; NMR refinement stuff
; Distance restraints type: No, Simple or Ensemble
disre = No
; Force weighting of pairs in one distance restraint: Conservative or Equal
disre-weighting = Conservative
; Use sqrt of the time averaged times the instantaneous violation
disre-mixed = no
disre-fc = 1000
disre-tau = 0
; Output frequency for pair distances to energy file
nstdisreout = 100
; Orientation restraints: No or Yes
orire = no
; Orientation restraints force constant and tau for time averaging
orire-fc = 0
orire-tau = 0
orire-fitgrp =
; Output frequency for trace(SD) and S to energy file
nstorireout = 100
; Free energy variables
free-energy = no
couple-moltype =
couple-lambda0 = vdw-q
couple-lambda1 = vdw-q
couple-intramol = no
init-lambda = -1
init-lambda-state = -1
delta-lambda = 0
nstdhdl = 50
fep-lambdas =
mass-lambdas =
coul-lambdas =
vdw-lambdas =
bonded-lambdas =
restraint-lambdas =
temperature-lambdas =
calc-lambda-neighbors = 1
init-lambda-weights =
dhdl-print-energy = no
sc-alpha = 0
sc-power = 1
sc-r-power = 6
sc-sigma = 0.3
sc-coul = no
separate-dhdl-file = yes
dhdl-derivatives = yes
dh_hist_size = 0
dh_hist_spacing = 0.1
; Non-equilibrium MD stuff
acc-grps =
accelerate =
freezegrps =
freezedim =
cos-acceleration = 0
deform =
; simulated tempering variables
simulated-tempering = no
simulated-tempering-scaling = geometric
sim-temp-low = 300
sim-temp-high = 300
; Ion/water position swapping for computational electrophysiology setups
; Swap positions along direction: no, X, Y, Z
swapcoords = no
adress = no
; User defined thingies
user1-grps =
user2-grps =
userint1 = 0
userint2 = 0
userint3 = 0
userint4 = 0
userreal1 = 0
userreal2 = 0
userreal3 = 0
userreal4 = 0
; Electric fields
; Format for electric-field-x, etc. is: four real variables:
; amplitude (V/nm), frequency omega (1/ps), time for the pulse peak (ps),
; and sigma (ps) width of the pulse. Omega = 0 means static field,
; sigma = 0 means no pulse, leaving the field to be a cosine function.
electric-field-x = 0 0 0 0
electric-field-y = 0 0 0 0
electric-field-z = 0 0 0 0
; Density guided simulation
density-guided-simulation-active = false
My peptide consists of Serine, Lysine, Glycine and Isoleucine beads :)
The bead types are in the .gro file here:
1GLY BB 1 5.500 5.970 8.457 0.0321 -0.1889 -0.1381
2ILE BB 2 5.539 6.248 8.433 -0.0441 0.1175 0.2417
2ILE SC1 3 5.291 6.428 8.382 0.0048 0.0187 -0.3530
3ILE BB 4 5.821 6.380 8.433 -0.5382 -0.0295 -0.2408
3ILE SC1 5 5.785 6.682 8.374 -0.1138 0.0255 -0.2124
4LYS BB 6 6.129 6.322 8.335 -0.4419 0.1299 0.1312
4LYS SC1 7 6.338 6.170 8.517 0.2437 -0.0474 -0.0635
4LYS SC2 8 6.401 5.981 8.708 0.0359 0.1261 0.1606
5SER BB 9 6.088 6.204 8.059 -0.1296 -0.0241 0.1312
5SER SC1 10 5.873 6.046 8.046 -0.2040 0.2811 0.1788
6ILE BB 11 6.413 6.154 7.931 -0.0365 0.0844 -0.0531
6ILE SC1 12 6.456 6.402 7.750 -0.0853 0.0600 -0.0976
7ILE BB 13 6.578 5.918 7.839 0.3101 0.1128 -0.0811
7ILE SC1 14 6.568 5.972 7.534 0.1234 -0.0627 -0.1056
8LYS BB 15 6.718 5.608 7.922 0.0321 0.0824 -0.1903
8LYS SC1 16 6.457 5.392 7.987 -0.0138 0.1463 -0.0142
8LYS SC2 17 6.332 5.296 8.139 0.0954 -0.1162 0.0266
9SER BB 18 6.992 5.441 7.970 -0.3956 0.3567 -0.2319
9SER SC1 19 6.961 5.203 7.877 0.0884 -0.0812 -0.0177
10ILE BB 20 7.279 5.491 8.193 0.1563 -0.2357 0.2483
10ILE SC1 21 7.486 5.322 8.037 -0.0443 -0.4676 0.2354
11ILE BB 22 7.386 5.781 8.482 0.1303 -0.1111 -0.3149
11ILE SC1 23 7.204 6.010 8.379 0.0793 -0.0047 0.0102
12LYS BB 24 7.265 5.625 8.796 0.0478 0.0462 -0.0177
12LYS SC1 25 7.082 5.399 8.693 0.2565 -0.0043 -0.2232
12LYS SC2 26 6.907 5.199 8.802 0.2128 0.2818 0.2625
13SER BB 27 7.105 5.634 9.109 -0.0574 -0.2658 -0.1664
13SER SC1 28 7.158 5.839 9.263 0.0154 0.1371 0.0659
14ILE BB 29 6.843 5.390 9.250 0.1102 0.0637 -0.1556
14ILE SC1 30 7.037 5.237 9.438 0.1397 0.1830 -0.0906
The .mdp out file is the example one suggested in the tutorials below:
define = -DPOSRES
; RUN CONTROL PARAMETERS
integrator = md
; Start time and timestep in ps
tinit = 0
dt = 0.001
nsteps = 125000
; For exact run continuation or redoing part of a run
init-step = 0
; Part index is updated automatically on checkpointing (keeps files separate)
simulation-part = 1
; mode for center of mass motion removal
comm_mode = linear
; number of steps for center of mass motion removal
nstcomm = 100
; group(s) for center of mass motion removal
comm_grps = Protein W_NA+_CL-
; LANGEVIN DYNAMICS OPTIONS
; Friction coefficient (amu/ps) and random seed
bd-fric = 0
ld-seed = -1
; ENERGY MINIMIZATION OPTIONS
; Force tolerance and initial step-size
emtol = 10
emstep = 0.01
; Max number of iterations in relax-shells
niter = 20
; Step size (ps^2) for minimization of flexible constraints
fcstep = 0
; Frequency of steepest descents steps when doing CG
nstcgsteep = 1000
nbfgscorr = 10
; TEST PARTICLE INSERTION OPTIONS
rtpi = 0.05
; OUTPUT CONTROL OPTIONS
; Output frequency for coords (x), velocities (v) and forces (f)
nstxout = 0
nstvout = 5000
nstfout = 5000
; Output frequency for energies to log file and energy file
nstlog = 1000
nstcalcenergy = 100
nstenergy = 1000
; Output frequency and precision for .xtc file
nstxout-compressed = 5000
compressed-x-precision = 1000
; This selects the subset of atoms for the compressed
; trajectory file. You can select multiple groups. By
; default, all atoms will be written.
compressed-x-grps =
; Selection of energy groups
energygrps =
; NEIGHBORSEARCHING PARAMETERS
; cut-off scheme (Verlet: particle based cut-offs)
cutoff-scheme = Verlet
; nblist update frequency
nstlist = 20
; Periodic boundary conditions: xyz, no, xy
pbc = xyz
periodic-molecules = no
; Allowed energy error due to the Verlet buffer in kJ/mol/ps per atom,
; a value of -1 means: use rlist
verlet-buffer-tolerance = 0.005
; nblist cut-off
rlist = 1.2
; long-range cut-off for switched potentials
; OPTIONS FOR ELECTROSTATICS AND VDW
; Method for doing electrostatics
coulombtype = PME
coulomb-modifier = Potential-shift-Verlet
rcoulomb-switch = 0
rcoulomb = 1.2
; Relative dielectric constant for the medium and the reaction field
epsilon-r = 1
epsilon-rf = 0
; Method for doing Van der Waals
vdwtype = Cut-off
vdw-modifier = Force-switch
; cut-off lengths
rvdw_switch = 1.0
rvdw = 1.2
; Apply long range dispersion corrections for Energy and Pressure
DispCorr = No
; Extension of the potential lookup tables beyond the cut-off
table-extension = 1
; Separate tables between energy group pairs
energygrp-table =
; Spacing for the PME/PPPM FFT grid
fourierspacing = 0.12
; FFT grid size, when a value is 0 fourierspacing will be used
fourier-nx = 0
fourier-ny = 0
fourier-nz = 0
; EWALD/PME/PPPM parameters
pme-order = 4
ewald-rtol = 1e-05
ewald-rtol-lj = 0.001
lj-pme-comb-rule = Geometric
ewald-geometry = 3d
epsilon-surface = 0
implicit-solvent = no
; OPTIONS FOR WEAK COUPLING ALGORITHMS
; Temperature coupling
tcoupl = V-rescale
nsttcouple = -1
nh-chain-length = 10
print-nose-hoover-chain-variables = no
; Groups to couple separately
tc_grps = Protein W_NA+_CL-
; Time constant (ps) and reference temperature (K)
tau_t = 1.0 1.0
ref_t = 303.15 303.15
; pressure coupling
pcoupl = Berendsen
pcoupltype = Isotropic
nstpcouple = -1
; Time constant (ps), compressibility (1/bar) and reference P (bar)
tau_p = 5.0
compressibility = 4.5e-5 4.5e-5
ref_p = 1.0 1.0
; Scaling of reference coordinates, No, All or COM
refcoord_scaling = com
; OPTIONS FOR QMMM calculations
QMMM = no
; Groups treated Quantum Mechanically
QMMM-grps =
; QM method
QMmethod =
; QMMM scheme
QMMMscheme = normal
; QM basisset
QMbasis =
; QM charge
QMcharge =
; QM multiplicity
QMmult =
; Surface Hopping
SH =
; CAS space options
CASorbitals =
CASelectrons =
SAon =
SAoff =
SAsteps =
; Scale factor for MM charges
MMChargeScaleFactor = 1
; SIMULATED ANNEALING
; Type of annealing for each temperature group (no/single/periodic)
annealing =
; Number of time points to use for specifying annealing in each group
annealing-npoints =
; List of times at the annealing points for each group
annealing-time =
; Temp. at each annealing point, for each group.
annealing-temp =
; GENERATE VELOCITIES FOR STARTUP RUN
gen-vel = no
gen-temp = 300
gen-seed = -1
; OPTIONS FOR BONDS
constraints = h-bonds
; Type of constraint algorithm
constraint_algorithm = LINCS
; Do not constrain the start configuration
continuation = yes
; Use successive overrelaxation to reduce the number of shake iterations
Shake-SOR = no
; Relative tolerance of shake
shake-tol = 0.0001
; Highest order in the expansion of the constraint coupling matrix
lincs-order = 4
; Number of iterations in the final step of LINCS. 1 is fine for
; normal simulations, but use 2 to conserve energy in NVE runs.
; For energy minimization with constraints it should be 4 to 8.
lincs-iter = 1
; Lincs will write a warning to the stderr if in one step a bond
; rotates over more degrees than
lincs-warnangle = 30
; Convert harmonic bonds to morse potentials
morse = no
; ENERGY GROUP EXCLUSIONS
; Pairs of energy groups for which all non-bonded interactions are excluded
energygrp-excl =
; WALLS
; Number of walls, type, atom types, densities and box-z scale factor for Ewald
nwall = 0
wall-type = 9-3
wall-r-linpot = -1
wall-atomtype =
wall-density =
wall-ewald-zfac = 3
; COM PULLING
pull = no
; AWH biasing
awh = no
; ENFORCED ROTATION
; Enforced rotation: No or Yes
rotation = no
; Group to display and/or manipulate in interactive MD session
IMD-group =
; NMR refinement stuff
; Distance restraints type: No, Simple or Ensemble
disre = No
; Force weighting of pairs in one distance restraint: Conservative or Equal
disre-weighting = Conservative
; Use sqrt of the time averaged times the instantaneous violation
disre-mixed = no
disre-fc = 1000
disre-tau = 0
; Output frequency for pair distances to energy file
nstdisreout = 100
; Orientation restraints: No or Yes
orire = no
; Orientation restraints force constant and tau for time averaging
orire-fc = 0
orire-tau = 0
orire-fitgrp =
; Output frequency for trace(SD) and S to energy file
nstorireout = 100
; Free energy variables
free-energy = no
couple-moltype =
couple-lambda0 = vdw-q
couple-lambda1 = vdw-q
couple-intramol = no
init-lambda = -1
init-lambda-state = -1
delta-lambda = 0
nstdhdl = 50
fep-lambdas =
mass-lambdas =
coul-lambdas =
vdw-lambdas =
bonded-lambdas =
restraint-lambdas =
temperature-lambdas =
calc-lambda-neighbors = 1
init-lambda-weights =
dhdl-print-energy = no
sc-alpha = 0
sc-power = 1
sc-r-power = 6
sc-sigma = 0.3
sc-coul = no
separate-dhdl-file = yes
dhdl-derivatives = yes
dh_hist_size = 0
dh_hist_spacing = 0.1
; Non-equilibrium MD stuff
acc-grps =
accelerate =
freezegrps =
freezedim =
cos-acceleration = 0
deform =
; simulated tempering variables
simulated-tempering = no
simulated-tempering-scaling = geometric
sim-temp-low = 300
sim-temp-high = 300
; Ion/water position swapping for computational electrophysiology setups
; Swap positions along direction: no, X, Y, Z
swapcoords = no
adress = no
; User defined thingies
user1-grps =
user2-grps =
userint1 = 0
userint2 = 0
userint3 = 0
userint4 = 0
userreal1 = 0
userreal2 = 0
userreal3 = 0
userreal4 = 0
; Electric fields
; Format for electric-field-x, etc. is: four real variables:
; amplitude (V/nm), frequency omega (1/ps), time for the pulse peak (ps),
; and sigma (ps) width of the pulse. Omega = 0 means static field,
; sigma = 0 means no pulse, leaving the field to be a cosine function.
electric-field-x = 0 0 0 0
electric-field-y = 0 0 0 0
electric-field-z = 0 0 0 0
; Density guided simulation
density-guided-simulation-active = false
Please Log in or Create an account to join the conversation.
- vainikka
-

- Offline
- Admin
Less
More
- Posts: 91
1 year 11 months ago #9471
by vainikka
Replied by vainikka on topic Counter ions clumping together during production run
Hi.
There seems to be quite a few oddities in the mdp options you are using.
The issue you are getting is from setting epsilon_r = 1. The standard with Martini is 15, which should resolve the issues you're getting.
You can get a standard Martini mdp file from:
cgmartini.nl/index.php/force-field-parameters/input-parameters
There seems to be quite a few oddities in the mdp options you are using.
The issue you are getting is from setting epsilon_r = 1. The standard with Martini is 15, which should resolve the issues you're getting.
You can get a standard Martini mdp file from:
cgmartini.nl/index.php/force-field-parameters/input-parameters
Please Log in or Create an account to join the conversation.
- bart
-

- Offline
- Admin
Less
More
- Posts: 98
1 year 11 months ago #9472
by bart
Replied by bart on topic Counter ions clumping together during production run
Indeed, also you are using PME which is not the standard. We usually use RF for electrostatics (also part if the default settings).
Please Log in or Create an account to join the conversation.
- Anna Stephens
-
 Topic Author
Topic Author
- Offline
- Fresh Boarder
Less
More
- Posts: 5
1 year 11 months ago #9473
by Anna Stephens
Replied by Anna Stephens on topic Counter ions clumping together during production run
Brilliant I see that now! Thanks so much for getting back to me!
Anna
Anna
Please Log in or Create an account to join the conversation.
Time to create page: 0.102 seconds