Caveolin-membrane
- Details
- Last Updated: Wednesday, 21 August 2013 11:23
Cholesterol Modulates the Structure, Binding Modes and Energetics of Caveolin-Membrane Interactions. Durba Sengupta, J. Phys. Chem. B, Just Accepted Manuscript, 2012. DOI: 10.1021/jp3077886
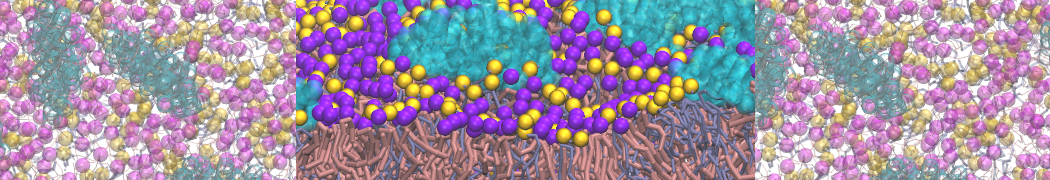
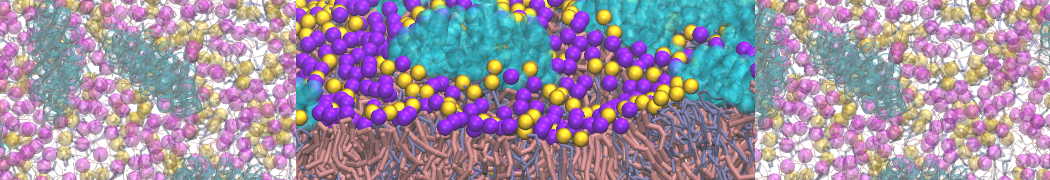
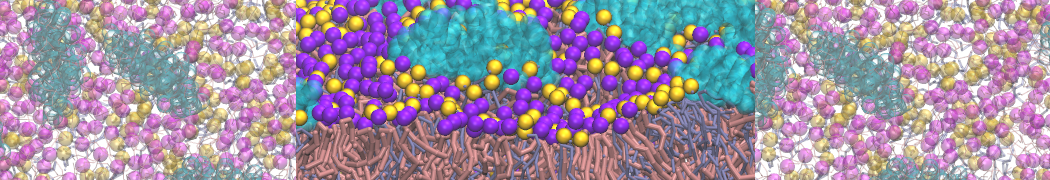
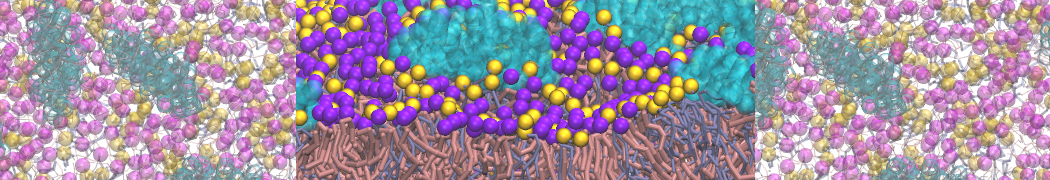
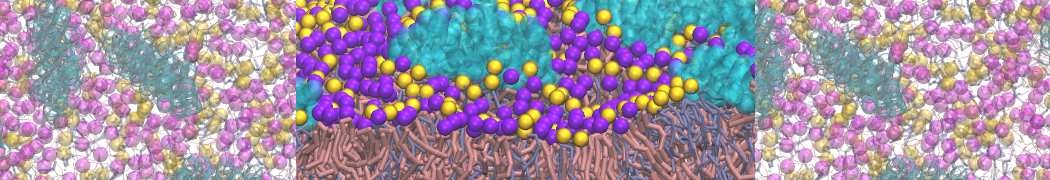
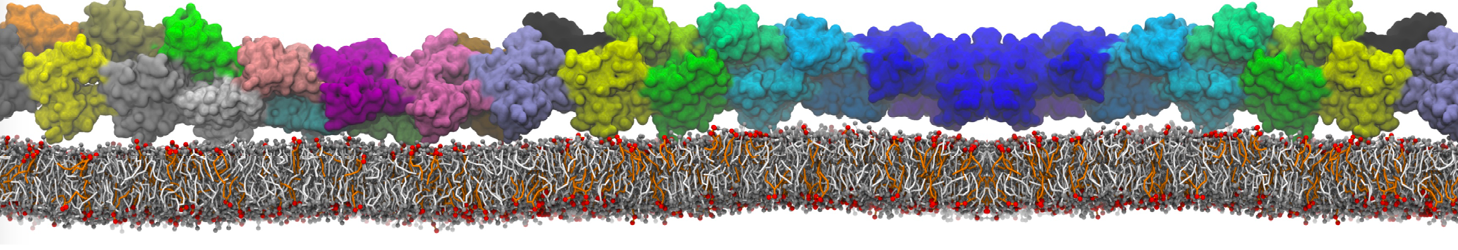
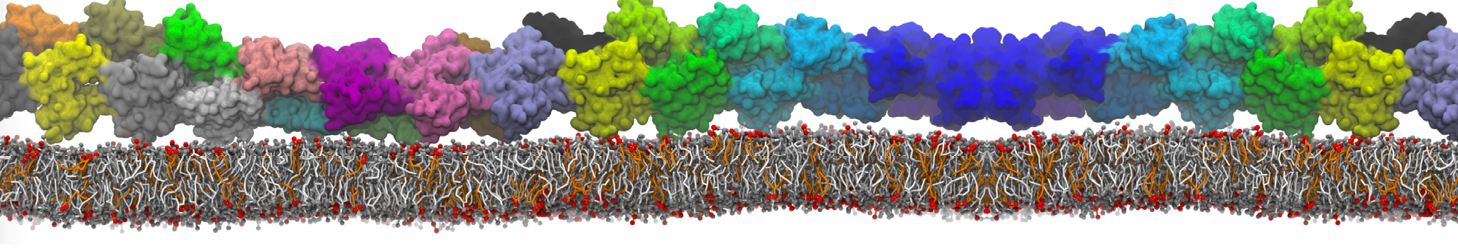
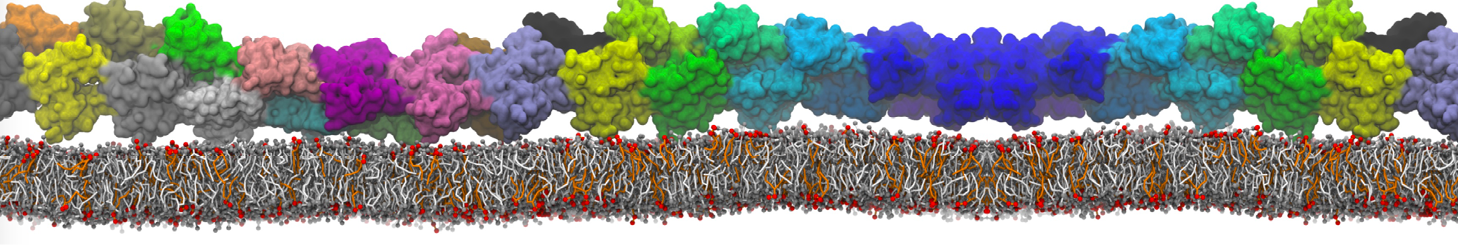
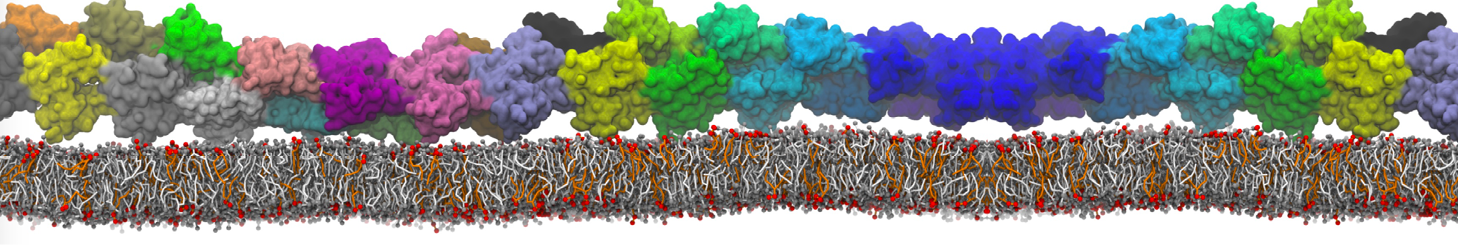
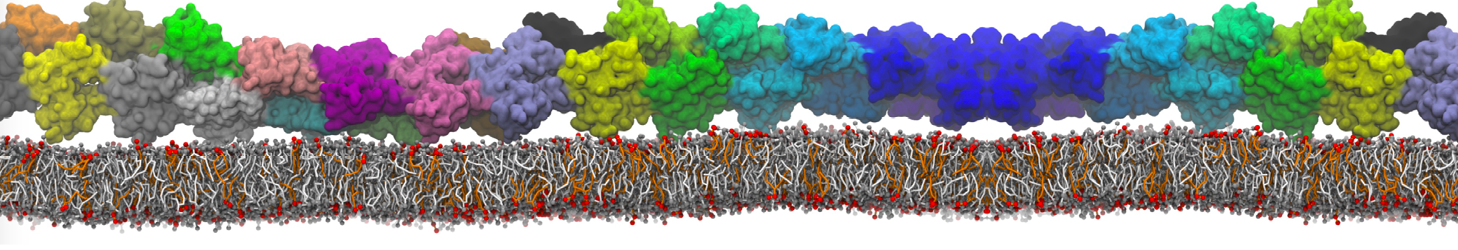
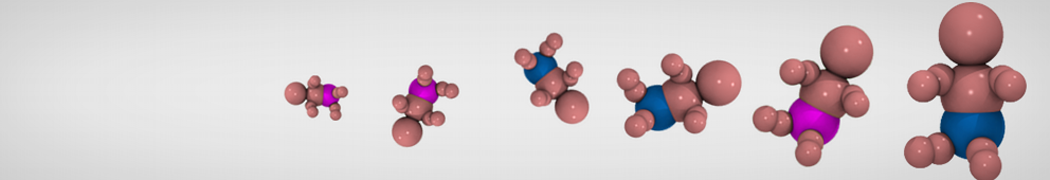
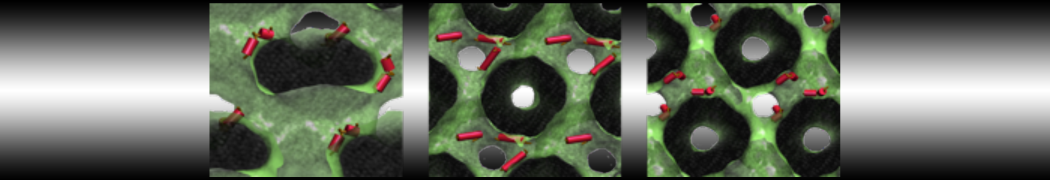
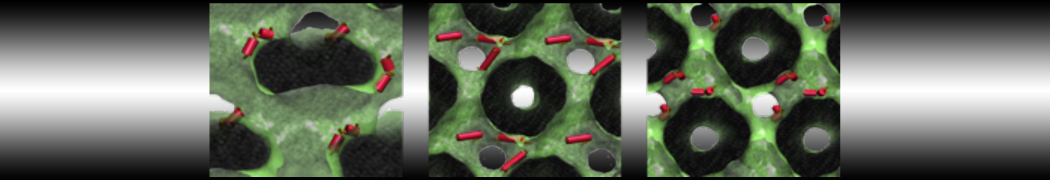
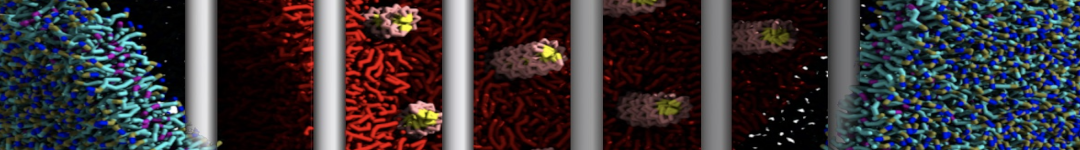
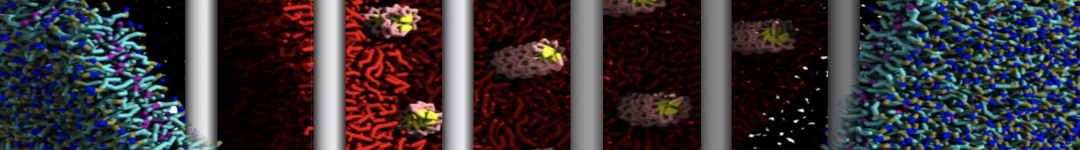
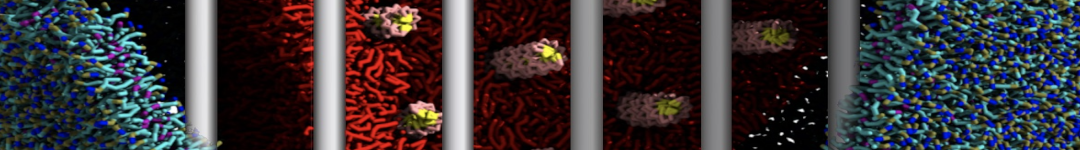
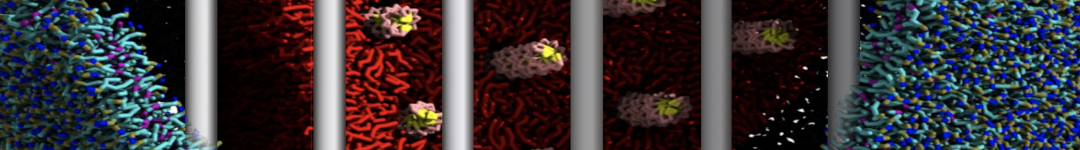
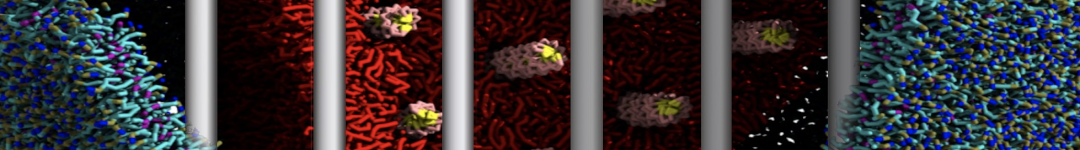
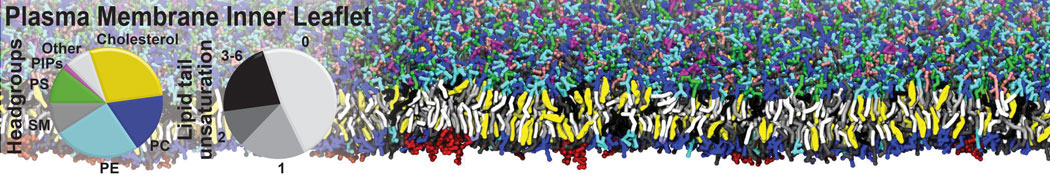
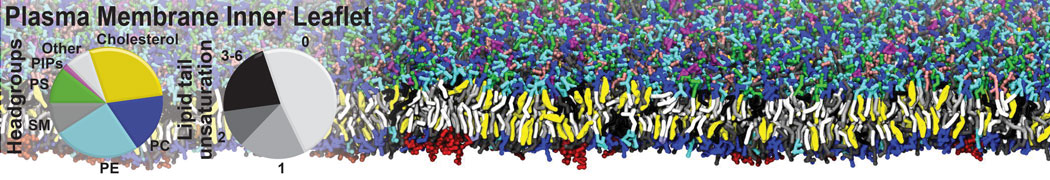
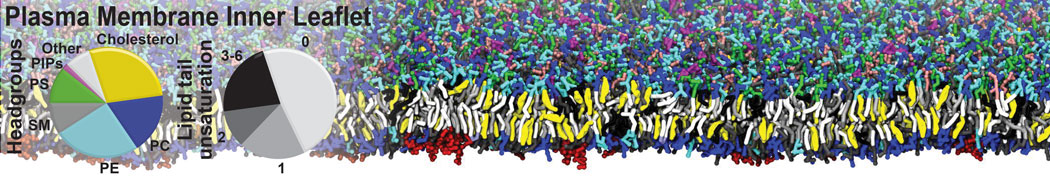
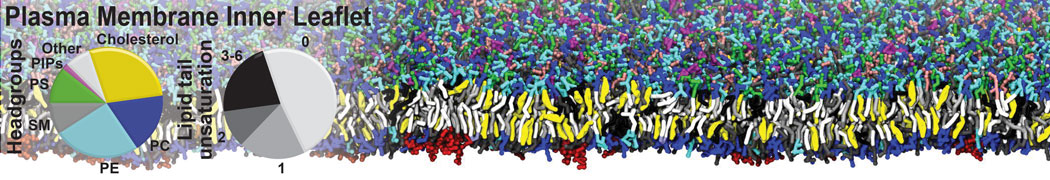
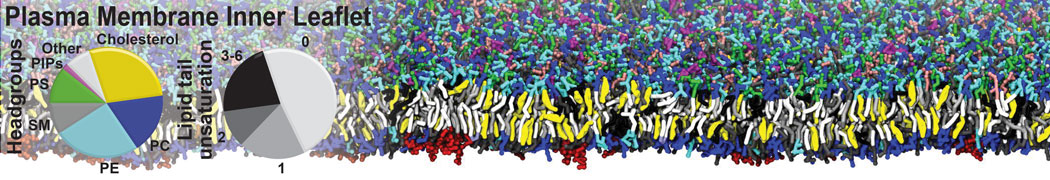
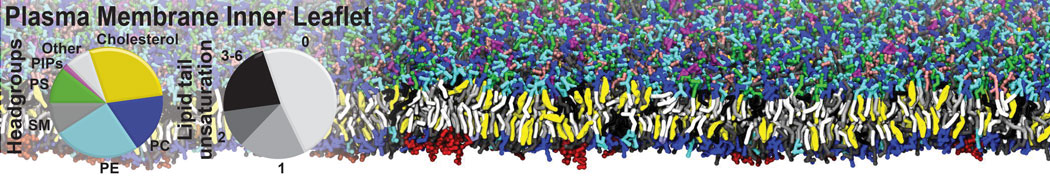
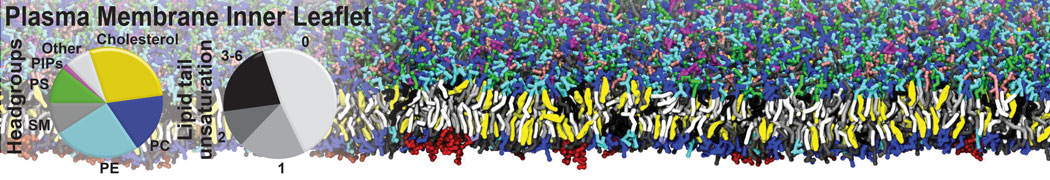
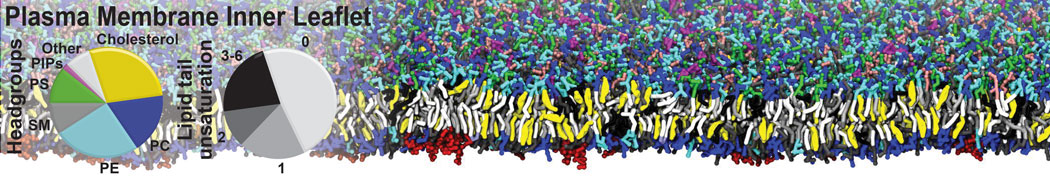
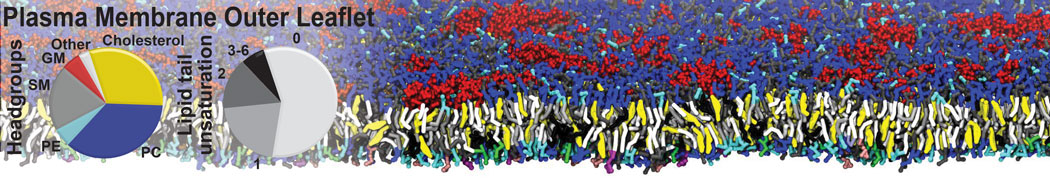
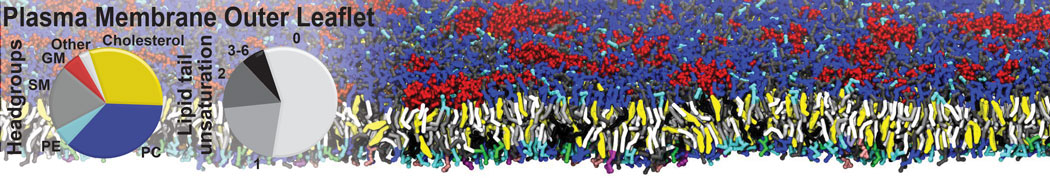
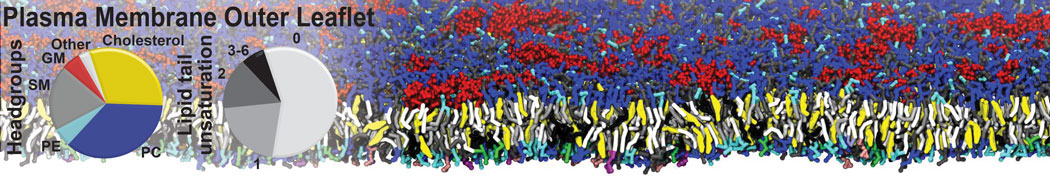
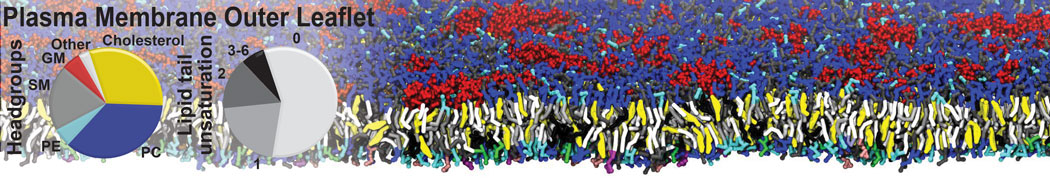
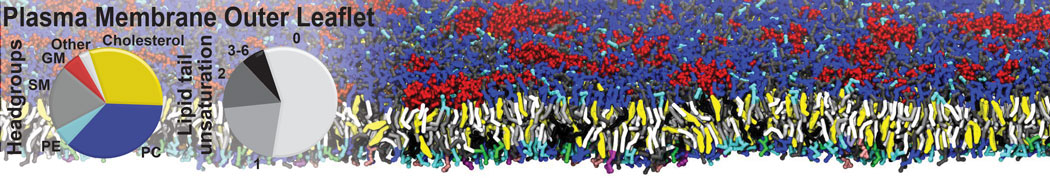
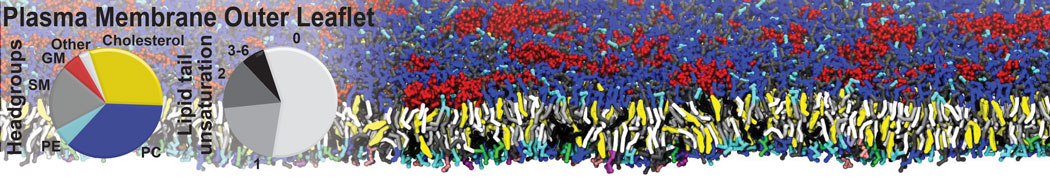
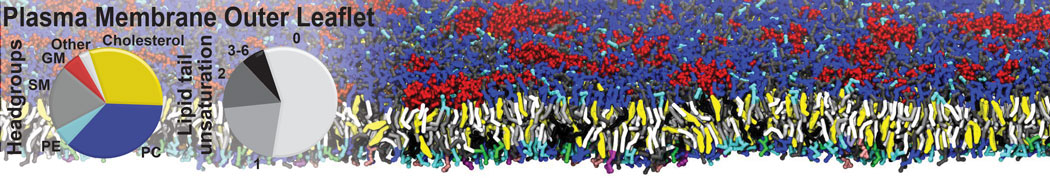
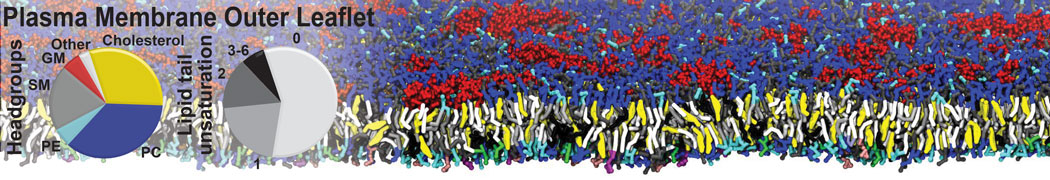
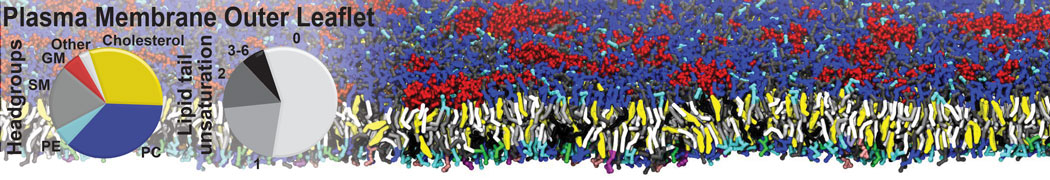
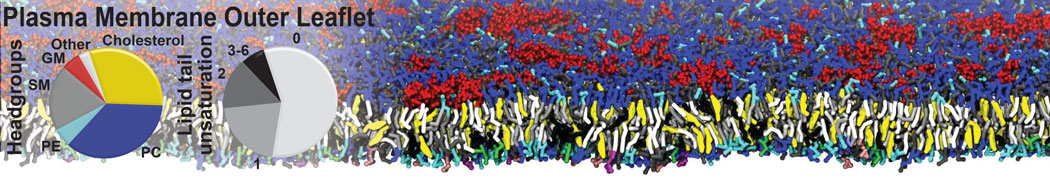
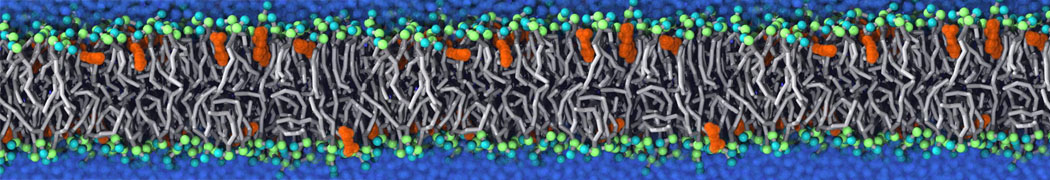
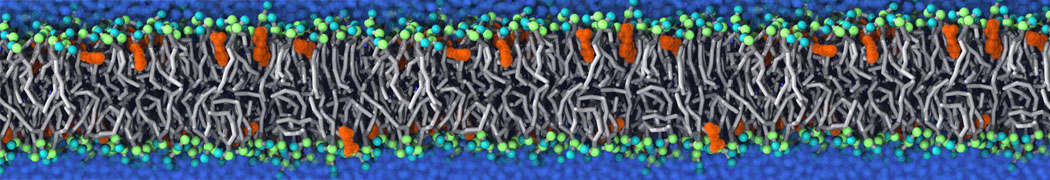
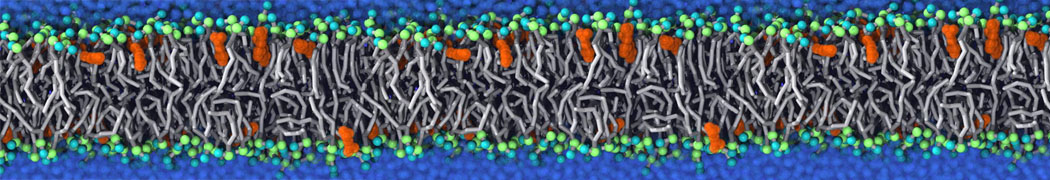
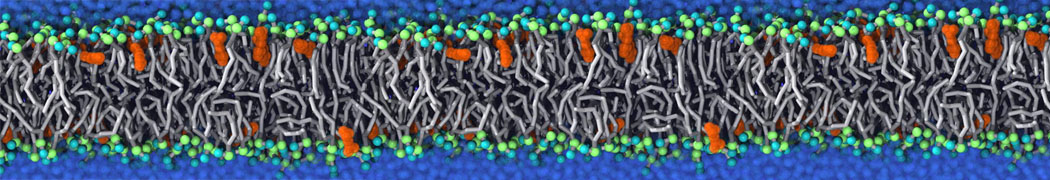
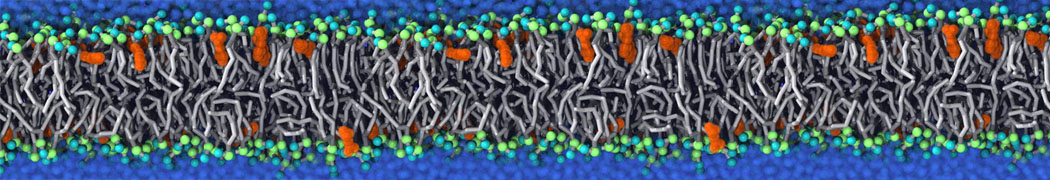
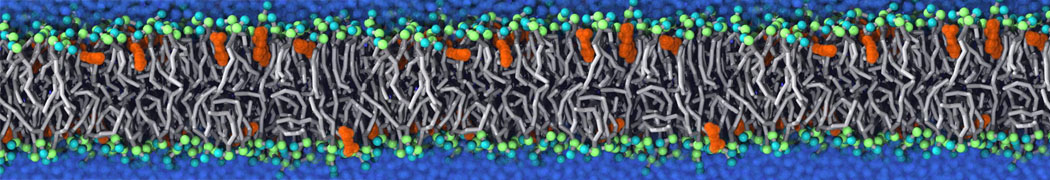
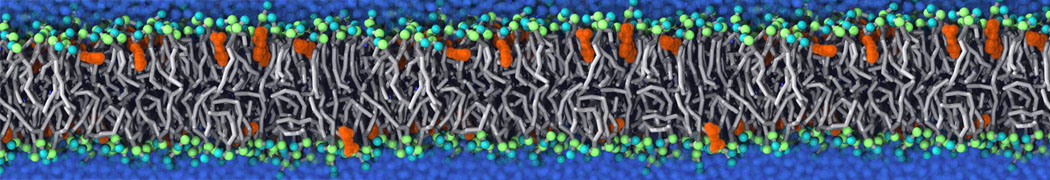
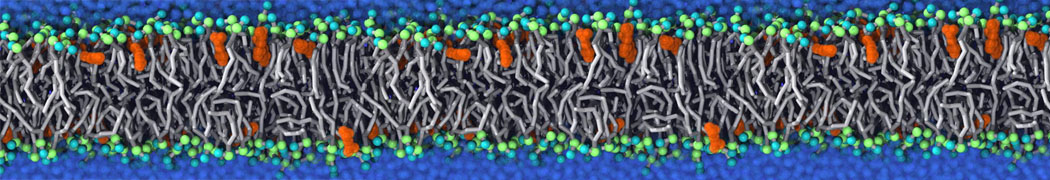
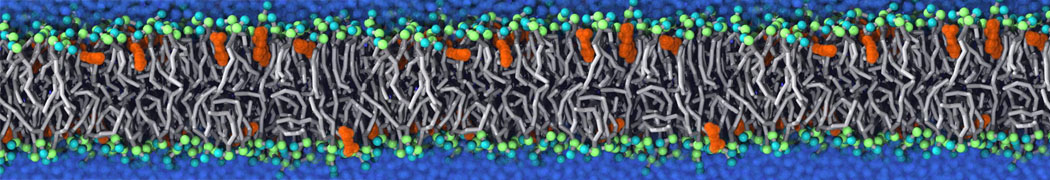
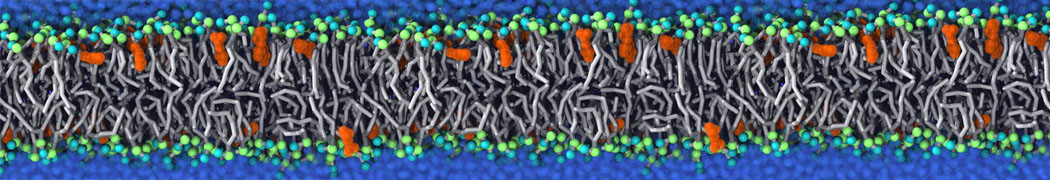
Caveolin-1 (cav-1) is an important membrane protein that plays a vital role in cellular signalling and trafficking by organising membrane domains. The peptide interacts with cholesterol-rich membranes and induces large morphological changes in them forming microdomains such as caveolae. Here, we use coarse-grained molecular dynamics simulations to study the interaction of cav-1 peptides with several model bilayer systems mimicking biological scenarios, such as cholesterol-rich domains, cholesterol-depleted domains and unsaturated lipid domains. We show that cholesterol modulates the depth as well as orientation of cav-1 binding to membranes. Furthermore, the presence of cholesterol establishes more open-conformations of cav-1, and we speculate that the binding modes and open-conformations could be responsible for inducing morphological changes in the bilayer. We also calculated the partitioning free energy to difference bilayers and show that binding to cholesterol-rich bilayers is more favorable than cholesterol depleted bilayers and the binding to unsaturated bilayers is the least favorable. Binding to cholesterol-rich bilayers also changes the pressure profile of the bilayer to which it is bound and thereby affects the local spontaneous curvature. Our results highlight molecular details of protein-lipid interplay and provide new insights into the effects of cav-1 in tuning the morphology of cholesterol rich membranes.

































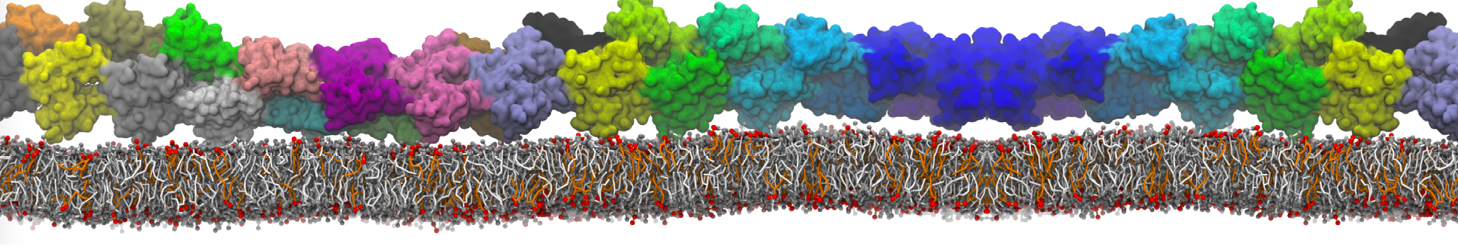










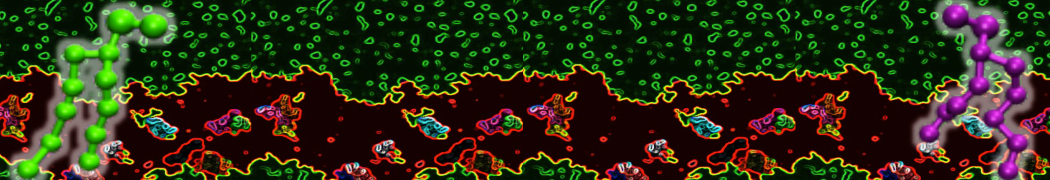
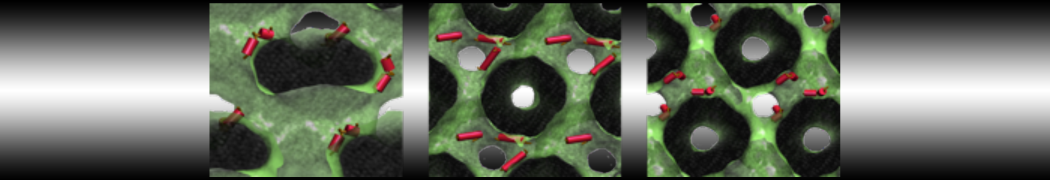
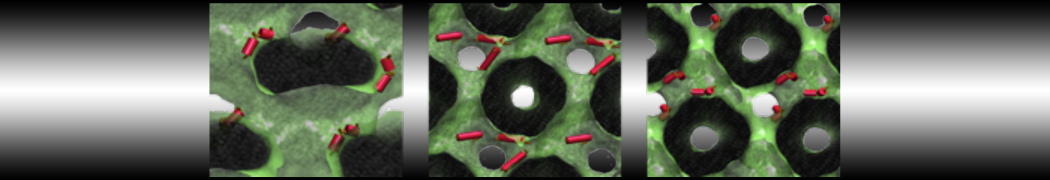
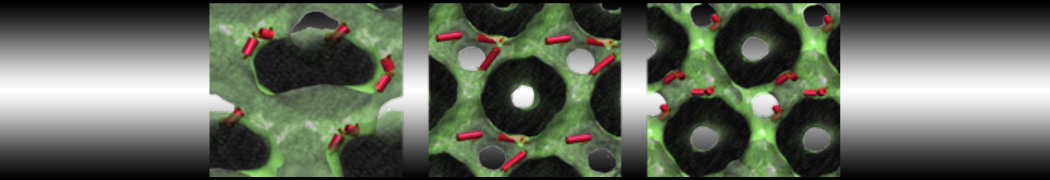
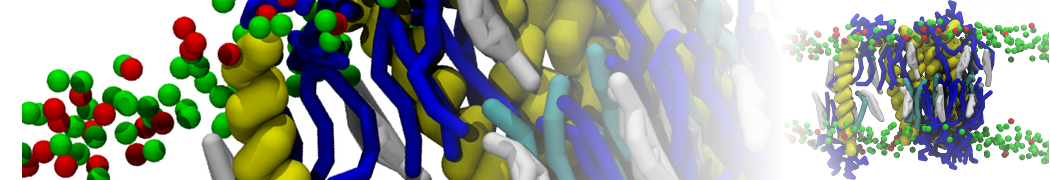
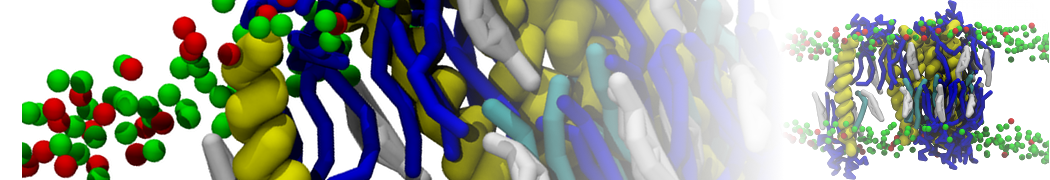
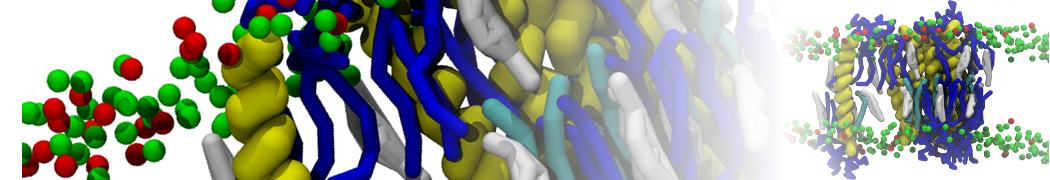
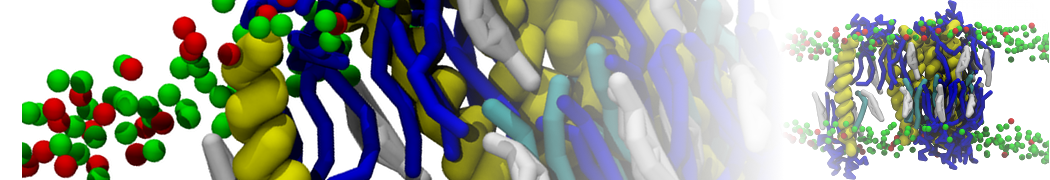
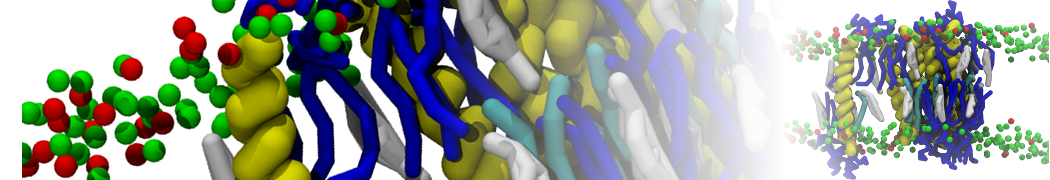
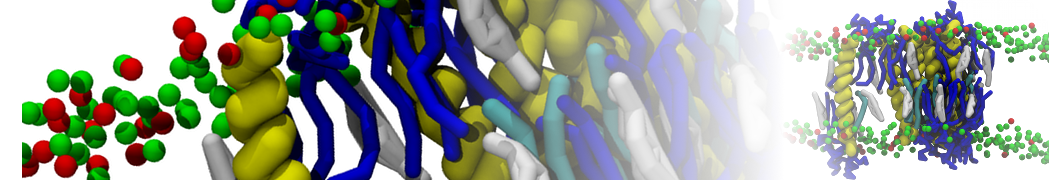
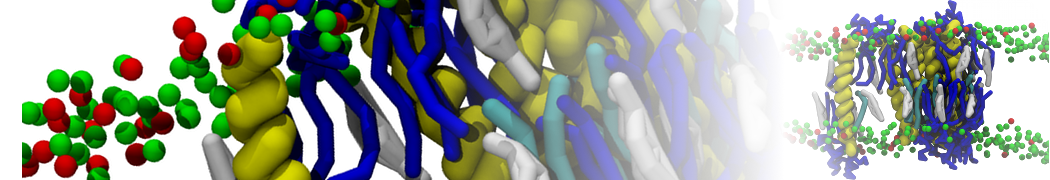
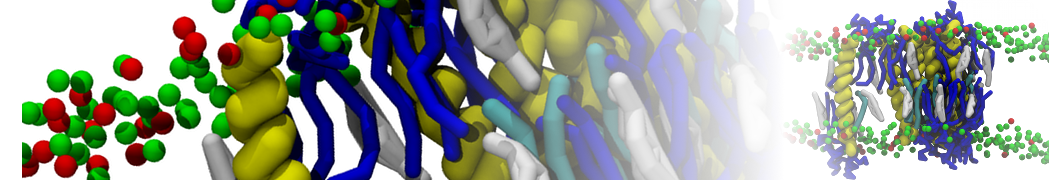
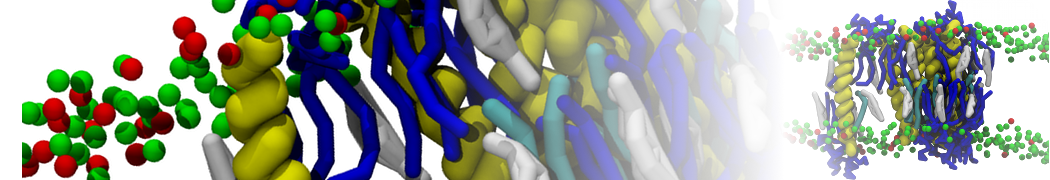
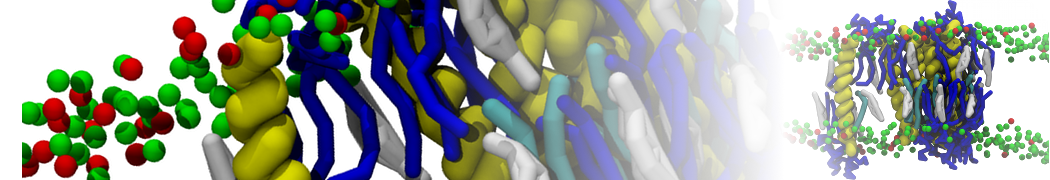
















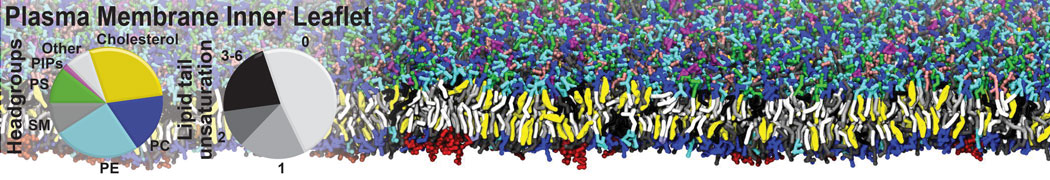
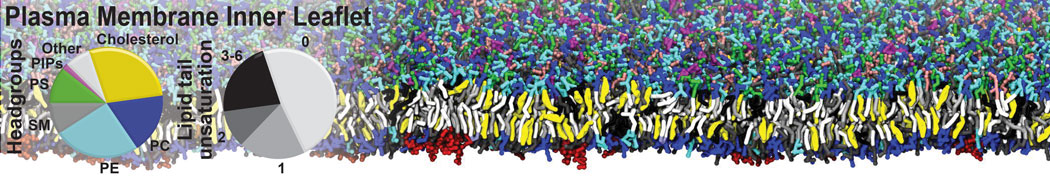
















 10° greater than that of GWALP23. The presence of R14 furthermore induces bilayer thinning and peptide displacement to “lift” the charged guanidinium toward the bilayer surface. By contrast, GWALP23-R12 exhibits multiple states that are in slow exchange on the NMR time scale, with CG-MD simulations indicating two distinct positions with different screw rotation angles in the membrane, along with an increased tendency to exit the lipid bilayer.
10° greater than that of GWALP23. The presence of R14 furthermore induces bilayer thinning and peptide displacement to “lift” the charged guanidinium toward the bilayer surface. By contrast, GWALP23-R12 exhibits multiple states that are in slow exchange on the NMR time scale, with CG-MD simulations indicating two distinct positions with different screw rotation angles in the membrane, along with an increased tendency to exit the lipid bilayer.